Translate this page into:
Neurophysiological pre-training protocols for performance and pain - a case report
*Corresponding author: Brogan Williams, Department of Research, College of Functional Movement Clinicians, Auckland, New Zealand. brogan.s.williams@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Lock A, Williams BS. Neurophysiological pre-training protocols for performance and pain - a case report. J Musculoskelet Surg Res. 2024;8:177-80. doi: 10.25259/JMSR_268_2023
Abstract
In the strength and conditioning domain, various methods are used to enhance range of motion (ROM), reduce pain, and enhance athletic performance. Among many popular interventions is the use of neurophysiological pre-training protocols, such as post-activation performance enhancement (PAPE). This case study aimed to detail the effects of a PAPE prescription and the accompanying neurophysiological mechanisms. While attempting to generate leg drive in a competition bench press position, a 35-year-old male powerlifter with 20 years of lifting experience complained of severe 9/10 right-sided hip pain. A gluteus medius dysfunction and abduction/extension intolerance were diagnosed. Using PAPE principles, a treatment strategy was designed targeting hip adduction to access hip extension better and restore appropriate ROM with leg drive. The participant reported 0/10 pain on the subsequent warm-up sets after the first six rounds of seated adductor isometric hold. The participant then performed a 220 kg 1 rep max bench press with an adequate ROM, leg drive, and no pain. More research should be done on neurophysiological rehabilitative strategies. This case study sheds light on the possibilities and calls for further research into the protocols and mechanisms discussed above.
Keywords
Biomechanics
Post-activation performance enhancement
Power
Powerlifting
Strength
INTRODUCTION
In the strength and conditioning domain, various methods are used to enhance range of motion (ROM), reduce pain, and enhance athletic performance. Among many popular interventions is the use of neurophysiological pre-training protocols – such as Post-Activation Performance Enhancement (PAPE), Proprioceptive Neuromuscular Facilitation, or Muscle Energy Techniques – which all involve physiological mechanisms such as Autogenetic and Reciprocal Inhibition (AI/RI), Atherogenic Neuromuscular Inhibition (ANI), and Post-Activation Potentiation (PAP).[1-4] This case study aimed to detail the effects of a PAPE prescription and the accompanying neurophysiological mechanisms on a 35-year-old male powerlifter.
CASE REPORT
While attempting to generate leg drive in a competition bench press position, a 35-year-old male powerlifter with 20 years of lifting experience complained of right-sided hip pain. The participant was 185 cm tall, weighed 104.3 g, and had no other health complications at this time. A visual pain scale (VAS) was used to determine the intensity of pain from 1 to 10, 1 being the least and 10 being the absolute worst pain. The pain was described as severe 8–9/10 deep hip pain along with rapid weakness/loss of hip and leg muscle contraction and inability to complete the attempt. An assessment was conducted immediately following the presentation of hip pain and was carried out in the gym. Upon further testing, certain movements were provocative. The pain was reproducible and aggravated with abduction, internal rotation, and hip extension accompanied by limited ROM. The ROM was tested through a functional movement screening, which required the participant to perform certain movements and motions to reveal the available range through visual inspection. The lack of ROM and pain at the hip negatively impacted the participant’s ability to set up and perform the bench press, making the position extremely painful and abolishing the necessary leg drive. The left-sided single-leg bridges were achievable; however, the right-sided were impossible due to the pain and weakness. The right-sided hip extension was reduced. The patient had no other unusual neurological activity or muscle weakness. A gluteus medius dysfunction and abduction/extension intolerance were hypothesized by the lead examiner.
Examiner information
A registered Australian Physiotherapist and rehabilitation specialist with over 30 years of experience was the lead examiner on this case study. The examiner’s assistant was a board-certified exercise physiologist with over 10 years of experience.
Interventions
Using PAPE principles, a treatment strategy was designed to target hip adduction to access hip extension better and restore appropriate ROM with leg drive. The participant was told to complete six sets of seated adduction maximum isometric holds against a foam roller between the knees for 10–20 s before preparing for the bench press [Figure 1]. The participant noted he could not last 10 s on the first attempt due to pain and disability. Once six quality sets were performed, the participant was instructed to set up as normal on the bench press but prioritize driving the knees inward into the bench, creating a strong adductor muscle activation coupled with hip extension. This setup technique was prescribed during the warm-up sets. The participant was told to complete two more sets of seated isometric holds before beginning actual working sets. After that, he was to resume using the standard bench technique and full-leg drive.
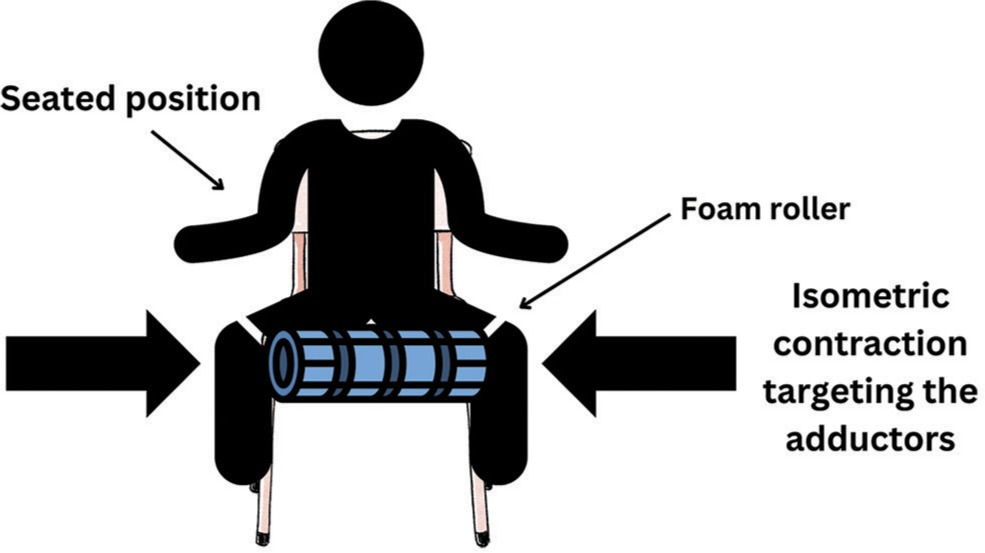
- An illustrated example of the exercise intervention.
RESULTS
The participant reported 0/10 pain on the subsequent bench press warm-up sets after the first six rounds of seated adductor isometric holds. The participant also achieved full ROM and measured it post-intervention. Following the bench press warm-up, the participant then performed a 220 kg 1 rep max bench press with adequate ROM, leg drive, and no pain.
DISCUSSION
A variety of neuropsychological interventions are used in strength sports to enhance performance. Like most sports, injuries can occur, and effective return-to-play strategies are required to aid the athlete through the rehabilitation process and back to peak performance. Neurological mechanisms can be advantageous in this pursuit, including PAPE, Proprioceptive Neuromuscular Facilitation, or Muscle Energy Techniques, which all involve physiological mechanisms such as AI/RI, ANI, and PAP. AI refers to a reflex arc from the Golgi Tendon Organ to the spinal cord on the stimulus of “tension” to the muscle’s 1b afferent fibers within the tendon. This stretch stimulus causes a reverse myotatic reflex, which inhibits the agonist muscle that is being targeted (the targeted muscle is the one being stretched or contracted).[3] RI refers to the decrease in neural activity (or drive) in the antagonist’s muscle due to voluntary contractions of the agonist’s muscle. This contraction stimulates the 1 a deep muscle spindles of the agonist muscle group, which detects muscle stretch (aka contraction) and “inhibits” the target muscle (the target muscle is NOT the one being stretched or contracted, but is affected through the muscle spindles).[3-5] Both Golgi tendon organ (AI) and muscle spindles (RI) mechanisms can be used to prepare an athlete for the specific task ahead and are active mechanisms with such interventions as proprioceptive neuromuscular facilitation or muscle energy techniques and PAPE.[3-6]
Research shows that pathology at the hip joint often directly impacts the biomechanics of the hip and other structures above or below it, altering muscle function and how force is distributed through the posterior kinetic chain.[7-9] ANI is a neurophysiological presynaptic inhibitory mechanism that acts on damaged, irritated, or painful joints surrounding musculature.[10] This can cause muscle atrophy, joint dysfunction, and altered biomechanics.[11] Emerging research shows the negative effects associated with hip pathology on gluteal muscle activation and, consequently, hip extension.[12] These findings have supported the hypothesis that joint damage can cause neurological flexion facilitation and extension inhibition. This issue must be resolved throughout the rehabilitation process to restore normal function, performance, and adequate ROM.[10]
The PAPE describes a neurophysiological phenomenon that causes an enhancement in movement quality based on the muscle’s contractile history moments prior.[2] This is not to be confused with PAP, which is a transient increase in muscle force due to altered calcium levels and phosphorylation of myosin.[13] PAP, AI, and RI are all neurophysiological mechanisms that can occur under the PAPE protocol, which is used as a much broader term for enhancing performance (strength, power, and speed).[14] With proper knowledge of applied biomechanics, anatomy, and physiology, this protocol can be utilized to prescribe specific exercises and movements to emphasize muscle and fiber orientations and boost cellular and motor contribution in preparation for the subsequent activity.[15] This fantastic physiological phenomenon can be used when prescribing movements for rehabilitation purposes – or as a strategy to modulate pain and improve performance by “priming” specific muscles before performing high-loaded movements. Various researches support the PAPE protocol, showing improvements in vertical jump performance, sprint times, running deceleration times, athletic power, and velocity.[14-18]
The mechanisms associated with PAPE, PAP, and RI were specifically used in this study’s exercise prescription. This intervention aimed to activate the adductor musculature and inhibit abduction activity (the adductor group is an effective hip extensor in the supine bench press position).[19] The pre-activation sets of maximal isometric adductor holds were intended to cause an RI of the hip abductors. This allowed the adductors; adductor longus, gracilis, adductor brevis, pectineus, and adductor magnus (both anterior and posterior heads) to access hip extension by inhibiting the antagonistic and dysfunctional abductors (all fibers of the gluteus medius, gluteus minimus, and the tensor fasciae latae).[20] This opened up a neurophysiological window for the subject to regain hip extension and achieve the best Bench Press setup for performance.
The hip extension is used during the competition bench press to aid in optimizing the lumbar extension and managing the transmission of ground reaction forces. As a result, an efficient axial skeletal arch is established. The femurs must be abducted to allow a person to lie supine with their feet on the floor due to the position of the supine body on the bench press. When the athlete is under load, he or she actively extends the hip while simultaneously abducting the femur. This is a highly effective method. The primary hip extensors are the gluteus maximus, the posterior head of the adductor magnus, and the hamstrings – with the gluteus maximus and adductor magnus having the greatest cross-sectional area of all the hip extensors.[21] In this anatomic position, the posterior head of the adductor magnus has the greatest moment arm to provide hip extension.[19] The primary adductor muscles have established leverage for hip adduction, averaging almost 6 cm. This leverage is available for the production of adduction torque from both femoralon-pelvic and pelvic-on-femoral perspectives.[22]
CONCLUSION
More research should be done on neurophysiological rehabilitative strategies, including quality randomized controlled trials, to advance this research further. This case study explored the possible benefits of PAPE, PAP, AI, and RI as a rehabilitative and performance-based intervention. The prescribed protocol was able to improve the participants’ pain and performance. The scope of this case report was extremely narrow, and the results cannot be extrapolated to an entire population. However, the positive outcomes displayed in this report highlight the need for further research on this topic.
AUTHORS’ CONTRIBUTIONS
Both authors were involved in the writing, reviewing, and editing of this paper. The lead author (AL) created the intervention and treated the patient. The second author (BSW) was heavily involved in the research and writing of the paper. Both authors have critically reviewed and approved the final draft and are responsible for the manuscript’s content and similarity index.
ETHICAL APPROVAL
The Institutional Review Board approval is not required.
DECLARATION OF PATIENT CONSENT
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient has given his consent for his images and other clinical information to be reported in the journal. The patient understands that his name and initials will not be published, and due efforts will be made to conceal his identity, but anonymity cannot be guaranteed.
USE OF ARTIFICIAL INTELLIGENCE (AI)-ASSISTED TECHNOLOGY FOR MANUSCRIPT PREPARATION
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
CONFLICTS OF INTEREST
There are no conflicting relationships or activities.
FINANCIAL SUPPORT AND SPONSORSHIP
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- Autogenetic inhibition of motoneurones by impulses in group Ia muscle spindle afferents. J Physiol. 1979;293:173-95.
- [CrossRef] [PubMed] [Google Scholar]
- Post-activation performance enhancement strategies in sport: A brief review for practitioners. Hum Move. 2021;22:101-9.
- [CrossRef] [Google Scholar]
- Proprioceptive neuromuscular facilitation (PNF): Its mechanisms and effects on range of motion and muscular function. J Hum Kinet. 2012;31:105-13.
- [CrossRef] [PubMed] [Google Scholar]
- Comparing the effect of stretching and muscle energy technique in the management of lower cross syndrome. Pak J Med Health Sci. 2022;16:31-3.
- [CrossRef] [Google Scholar]
- The role of muscle proprioceptors in proprioceptive neuromuscular facilitation (PNF) stretching. 2019. Available from: https://advance.muschealth.org/library/2019/september/pnf-stretching [Last accessed on 2023 Mar 01]
- [Google Scholar]
- The effectiveness of PNF versus static stretching on increasing hip-flexion range of motion. J Sport Rehabil. 2018;27:289-94.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical biomechanics of the hip joint. Hip Arthrosc Hip Joint Preserv Surg. 2014;9:1-19.
- [CrossRef] [Google Scholar]
- Effects of low back pain on the relationship between the movements of the lumbar spine and hip. Hum Mov Sci. 2004;23:21-34.
- [CrossRef] [PubMed] [Google Scholar]
- Hip and lumbar spine physical examination findings in people presenting with low back pain, with or without lower extremity pain. J Orthop Sports Phys Ther. 2017;47:163-72.
- [CrossRef] [PubMed] [Google Scholar]
- Hip muscle inhibition after hip arthroscopy: A role for neuromuscular electrical stimulation. Int J Sports Phys Ther. 2020;15:1222-8.
- [CrossRef] [PubMed] [Google Scholar]
- Arthrogenic muscle inhibition: A limiting factor in joint rehabilitation. J Sport Rehabil. 2000;9:135-59.
- [CrossRef] [Google Scholar]
- Arthrogenic neuromusculature inhibition: A foundational investigation of existence in the hip joint. Clin Biomech. 2013;28:171-7.
- [CrossRef] [PubMed] [Google Scholar]
- Postactivation potentiation: An introduction. Int J Sports Phys Ther. 2011;6:234-40.
- [Google Scholar]
- Postactivation performance enhancement (PAPE) using a vertical jump to improve Vertical Jump Performance. J Sports Med Phys Fitness. 2022;62:1419-26.
- [CrossRef] [PubMed] [Google Scholar]
- Time to differentiate postactivation “potentiation” from “performance enhancement” in the strength and conditioning community. Sports Med. 2020;50:1559-65.
- [CrossRef] [PubMed] [Google Scholar]
- Post-activation performance enhancement (PAPE) after a single-bout of high-intensity flywheel resistance training. Biol Sport. 2020;37:343-50.
- [CrossRef] [PubMed] [Google Scholar]
- Does one heavy load back squat set lead to postactivation performance enhancement of three-point explosion and sprint in third division American football players? BMC Sports Sci Med Rehabil. 2021;13:64.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of post-activation performance enhancement (PAPE) induced by a plyometric protocol on deceleration performance. J Hum Kinet. 2021;80:5-16.
- [CrossRef] [PubMed] [Google Scholar]
- Kinesiology of the hip: A focus on muscular actions. J Orthop Sports Phys Ther. 2010;40:82-94.
- [CrossRef] [PubMed] [Google Scholar]
- Anatomy of the abductor muscles of the hip as studied by computed tomography. J Bone Joint Surg. 1987;69:1021-31.
- [CrossRef] [Google Scholar]
- Biomechanics and motor control of human movement. Hoboken, NJ: Wiley; 2009.
- [CrossRef] [Google Scholar]






