Translate this page into:
Sternoclavicular joint septic arthritis and osteomyelitis
Corresponding Author:
Ali M Al Ahmari
King Abdullah Medical City, Makkah
Saudi Arabia
alahmari.a@kamc.med.sa
| How to cite this article: Al Ahmari AM, Alanis IM. Sternoclavicular joint septic arthritis and osteomyelitis. J Musculoskelet Surg Res 2019;3:363-366 |
Abstract
Septic arthritis is an emergency condition in orthopedics because any delay in diagnosis and treatment can lead to the development of osteomyelitis and permanent damage to the bones and articulating surfaces. Septic arthritis of the sternoclavicular joint (SCJ) is an especially unusual infection and can be associated with serious complications such as osteomyelitis, chest wall abscess, and mediastinitis. Early diagnosis and prompt management are keys for avoiding the development of untoward complications. The aim of this report is to present our management of a case with no well-known cause for the presence of infection in SCJ and to compare the findings to the literature. This case report describes the course of a patient with medical comorbidities (hypertension, ischemic heart disease, and diabetes mellitus) who was managed surgically for SCJ septic arthritis.Introduction
The sternoclavicular joint (SCJ) is a rare site for septic arthritis. This type of sepsis is rare (0.5%–1.0% of all joint infections), and its cause is unknown in 23% of patients.[1],[2] Some risk factors identified in previous studies include intravenous (IV) drug use, distal infection, diabetes mellitus (DM), trauma, and an infected central venous line.[1],[2] Imaging modalities such as computed tomography (CT) and magnetic resonance imaging (MRI) can determine the severity of the infection.[3],[4]
Complications such as osteomyelitis,[5] large abscess formation,[6],[7] mediastinitis,[8] and empyema[9] can occur if the treatment of septic arthritis is delayed. Surgical debridement is urgent in septic arthritis and should be supported by suitable antibiotics to decrease the risk of irreversible tissue damage and potential mortality.[10],[11]
A scarce amount of literature is available on this topic, but previous studies have discussed the presentations, causes, and case management options.[12],[13],[14] The aim of this report is to present our management in a case with no well-known cause for the presence of infection in SCJ and to compare the findings to available literature.
Case Report
Our case report involved a 66-year-old Saudi male patient. He had been diagnosed previously with hypertension, ischemic heart disease, and DM and was controlled with regular medication use. There was a history of percutaneous coronary intervention (PCI) 4 months before presentation at our hospital and he had been taking Plavix and aspirin since that time.
His symptoms started 1 week before presentation at which time he developed right shoulder pain and a decrease in the range of motion while at home. He first presented to a local hospital but denied the suggested admission to complete assessment and examinations. Rather, the patient left against medical advice with prescribed antibiotics. Four days later, he developed a high-grade fever with chills and rigors and finally presented to our emergency department (ED).
In the ED, the patient was observed by a multidisciplinary team for evaluation, diagnosis, and management. He was initially seen by the ED and internal medicine physicians followed by a cardiologist and thoracic and orthopedic surgeons. He presented with right shoulder pain and a limited range of motion as well as high-grade fever, tachycardia, and tachypnea. On further inquiry, there was no history of trauma to the right shoulder, anorexia, weight loss, or night sweats, and the patient also denied any history of dental or surgical procedures in the right upper limb, head, and neck. The examination showed some redness and fullness around the right SCJ, which was warm and tender upon palpation. Examination of the right glenohumeral joint was unremarkable. Baseline blood investigations, chest radiographs, and electrocardiography were done. An elevated white blood cell (WBC) count and positive procalcitonin levels were detected. The cardiologist ruled out a cardiac event. An ultrasound [Figure - 1] was requested and revealed right subpectoral muscle fluid collection inferior to the SCJ. To rule out fluid leakage into the chest cavity and mediastinum, a chest CT scan [Figure - 2] was suggested by the thoracic surgeon. The CT scan showed no indication of fluid leakage into the chest cavity.
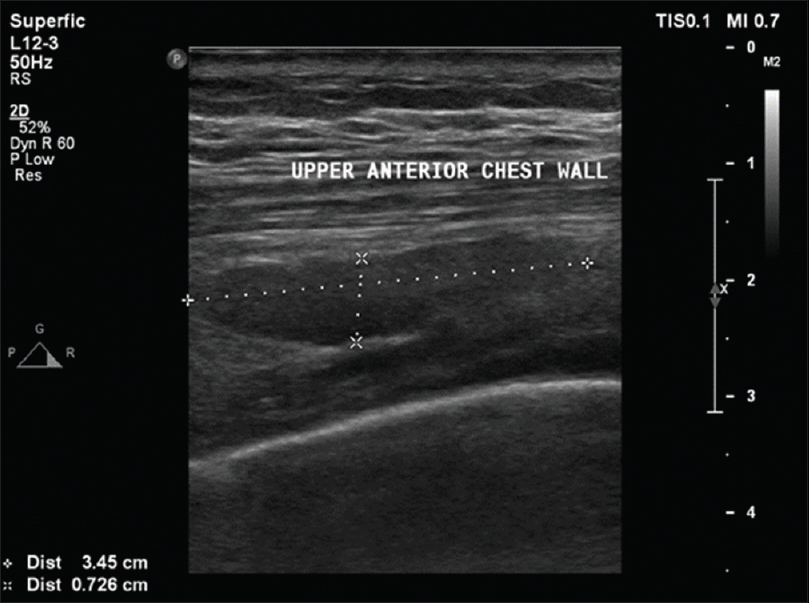 |
| Figure 1: The chest ultrasound showed the right subpectoral muscle fluid collection inferior to the sternoclavicular joint |
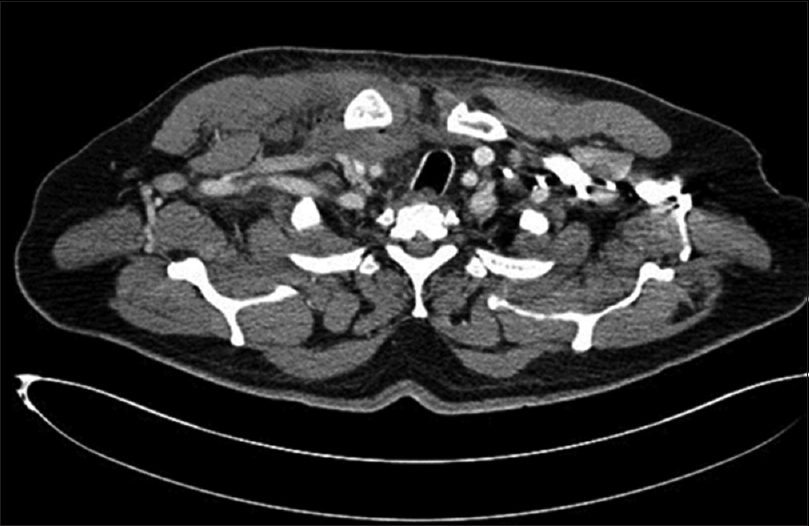 |
| Figure 2: Computed tomography scan of the chest showed no fluid leakage into the chest cavity |
The detailed assessment of the chest included MRI with contrast. It showed a chest wall abscess [Figure - 3]a measuring 11 cm × 4 cm surrounding the SCJ extending posteriorly. The abscess was located at the junction of the right subclavian and internal jugular veins and had displaced the trachea to the left side. Osteomyelitis [Figure - 3]b of the medial end of the clavicle was also detected. A provisional diagnosis of SCJ septic arthritis was hence confirmed radiographically.
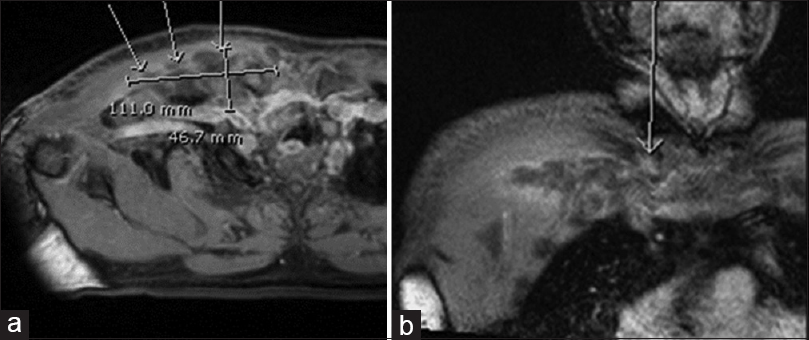 |
| Figure 3: (a) Axial section of the chest magnetic resonance imaging showing a chest wall abscess measuring 11 cm × 4 cm. (b) Coronal section of the chest magnetic resonance imaging; arrow points to osteomyelitis of the clavicle's medial end |
We planned for emergency surgery (same day of admission) considering the patient's condition and findings. The patient was positioned in a supine position with a roll of sheets in between the shoulders. A hockey-stick incision was used to expose the SCJ. The short arm was located over the medial part of the clavicle, and the long arm was parallel to the lateral side of the sternum over the right SCJ. The head of the clavicle and the body of the manubrium were exposed. A protective osteotomy was performed to the medial 2 cm of the right clavicle, and debridement was carried out to the surrounding tissues and along the downward side of the surgical wound to drain the pus pocket.
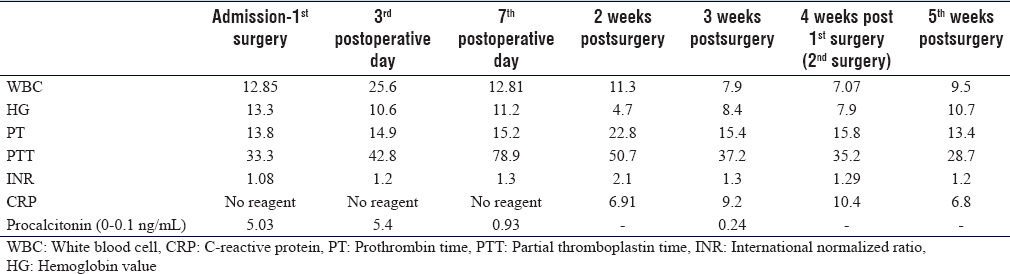
The WBC count was high upon admission, but it decreased significantly after treatment. Intraoperative wound cultures were sent for analysis. The C-reactive protein (CRP) was requested from the admission date, but unfortunately, there was no reagent until 2 weeks after the 1st operation. The result was 6.9 mg/L, and it increased gradually after the 2nd operation (4 weeks after the first surgery). The CRP then decreased gradually up to 2.6 mg/L on discharge [Table - 1]. Bacterial cultures were positive for Staphylococcus aureus. No growth was seen on fungal or tuberculosis cultures.
Intravenous antibiotics were initiated based on sensitivity testing. Clindamycin was started on admission followed by IV cloxacillin (based on the culture and sensitivity report) for approximately 4 weeks. This was followed by a switch to IV cefazolin for 2 weeks (based on infectious disease specialist advice) and then to oral cefuroxime (at discharge) for 2 more weeks.
The patient was moved to the intensive care unit after surgery as recommended by the anesthetists because of his recent PCI. The patient was moved to the general ward on the 5th postoperative day. The patient showed continuous improvement in his symptoms and became afebrile and pain-free, followed by a lack of tenderness in the right SCJ with pain-free shoulder movements. His lab results improved, but there was continuous discharge along the inferior end of the parasternal region of the incision. Thus, we opted for surgical debridement and irrigation (2nd surgery). Daily wound dressing changes were done in addition to IV antibiotics, leading to a healed wound within a few days.
The patient also complained of low back pain during the course of treatment. On further inquiry, he reported that the pain began 2 weeks before the right shoulder pain. There was no history of weakness or paresthesia in his lower limbs, and he had an intact bladder and normal bowel control. Radiographs of the lumbar spine showed involvement of the L3 and L4 vertebrae. A lumbosacral MRI [Figure - 4] showed enhancement of the L3 and L4 vertebral bodies and intervertebral discs that are associated with paravertebral and epidural soft-tissue enhancement. These findings suggested pyogenic/granulomatous spondylodiscitis.
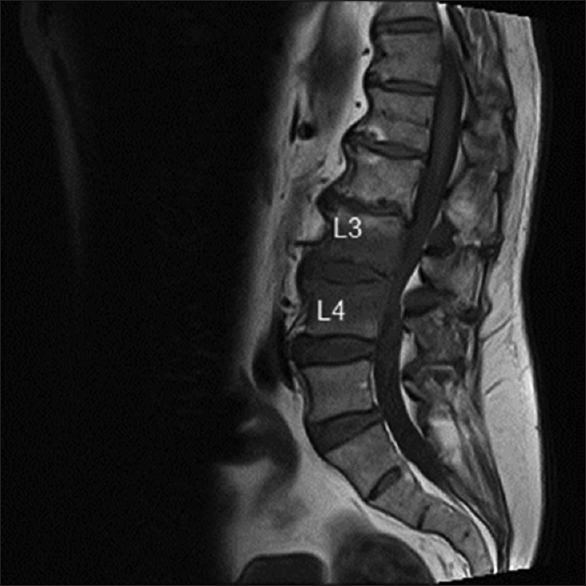 |
| Figure 4: Magnetic resonance imaging of the lumbosacral spine showed enhancement of the L3 and L4 vertebral bodies and intervertebral discs with associated paravertebral and epidural soft-tissue enhancement |
An interventional radiologist biopsied the lumbar lesion, but the result was inconclusive. Spinal surgeons were consulted, and they decided to perform an open biopsy to isolate the cause of infection; however, the patient refused to undergo the procedure; thus, we found no correlation between his diagnosis and the lower back pain.
The patient completed his treatment and was discharged with an acceptable range of right shoulder motion, laboratory findings within normal ranges, and on oral antibiotics as per the infectious disease department's protocol. It has been 20 months since his discharge. The patient's back pain did not improve, and he was found to be relapse-free at his recent follow-up in our outpatient department.
Discussion
SCJ septic arthritis is rare, but it can lead to serious complications such as osteomyelitis, chest wall abscess, mediastinitis, or myositis – all of these have a heightened risk of irreversible tissue damage and likely death.[2],[6] SCJ usually presents with chest pain localizing to the SCJ (78%), fever (65%), shoulder pain (24%), and rare neck pain (2%).[1],[15] Our patient presented with fever and pain right shoulder. This case presented with arthritis in addition to proximal-end right clavicle osteomyelitis with a short presentation period and no defined cause.
Ross and Shamsuddin identified certain risk factors such as IV drug use (21%), distant site of infection (15%), DM (13%), trauma (12%), and an infected central venous line (9%); however, 23% of the patients in their case series (n = 180 cases) had none of these risk factors.[1] Our patient had none of these except DM, which was controlled by medication. Therefore, our findings agree with previous studies in which the risk might be DM.
He had undergone surgery 4 months before presentation at our hospital. The PCI might have exposed him to elevated central venous line pressure and is one of the risks for the development of septic arthritis, leading to osteomyelitis and chest wall abscess. The patient had no information regarding the presence or absence of that intervention. He also was not exposed to any other risks that were cleared in history. There is a remote possibility that he already had developed a subclinical infection that led slowly to the chest wall abscess and osteomyelitis by the time he contacted the medical facility in his city with fever and shoulder pain.
The presence of chronic back pain 2 weeks before shoulder pain onset and fever development indicated the possibility of lumbar discitis but did not appear to be related to our case's presentation. However, failure of the CT-guided biopsy to detect the infecting organism and the patient's refusal to undergo an open biopsy limited our ability to help the patient complete his treatment. His age and the presence of DM alone were not enough to suggest multifocal bone infection.
The rate of positive cultures was 36% after surgical debridement in previous reports, and S. aureus was responsible for half of these infections.[2],[8] In our case, the same organism was isolated after analyzing the deep wound culture and was treated accordingly. In addition, our patient developed a chest wall abscess and osteomyelitis-related complications similar to those observed and documented in previous case studies.[6],[7] Case management with a multidisciplinary approach yielded the best outcome. The liaison of orthopedic, spinal, and thoracic surgeons, as well as the infectious disease and cardiology departments, helped to rule out and manage any relevant issues, especially comorbidities such as DM, by monitoring and regulating blood pressure, which played an important role in wound management.
The role of radiology was instrumental in this diagnosis because the ultrasound revealed pus collection, and the CT scan and MRI provided additional details such as bone osteomyelitis. The extension and boundaries of the collection helped rule out leakage into the thoracic cavity. SCJ septic arthritis is rare; cases with no apparent cause of presentation should be treated as an orthopedic emergency.
Conclusions
Septic joint arthritis is an emergency that calls for aggressive steps by a multidisciplinary team to avoid serious and life-threatening complications. Septic SCJ arthritis remains rare. Cases without a defined cause and multiple medical comorbidities (such as ours) call for a deeper investigation. Maintaining a high index of doubt can lead to a timely and prompt diagnosis. The shorter period of presentation further complicates the previously developed complications such as chest wall abscess and osteomyelitis in the right clavicle.
Ethical consideration
Approval was obtained.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Authors' contributions
All authors shared in data collection and analysis. All authors wrote the initial draft and critically reviewed and approved the final draft. They are responsible for the content and similarity index of the manuscript.
| 1. | Ross JJ, Shamsuddin H. Sternoclavicular septic arthritis: Review of 180 cases. Medicine (Baltimore) 2004;83:139-48. [Google Scholar] |
| 2. | Tanaka Y, Kato H, Shirai K, Nakajima Y, Yamada N, Okada H, et al. Sternoclavicular joint septic arthritis with chest wall abscess in a healthy adult: A case report. J Med Case Rep 2016;10:69. [Google Scholar] |
| 3. | Burkhart HM, Deschamps C, Allen MS, Nichols FC 3rd, Miller DL, Pairolero PC. Surgical management of sternoclavicular joint infections. J Thorac Cardiovasc Surg 2003;125:945-9. [Google Scholar] |
| 4. | Song HK, Guy TS, Kaiser LR, Shrager JB. Current presentation and optimal surgical management of sternoclavicular joint infections. Ann Thorac Surg 2002;73:427-31. [Google Scholar] |
| 5. | Tecce PM, Fishman EK. Spiral CT with multiplanar reconstruction in the diagnosis of sternoclavicular osteomyelitis. Skeletal Radiol 1995;24:275-81. [Google Scholar] |
| 6. | Wohlgethan JR, Newberg AH, Reed JI. The risk of abscess from sternoclavicular septic arthritis. J Rheumatol 1988;15:1302-6. [Google Scholar] |
| 7. | Van Linthoudt D, Velan F, Ott H. Abscess formation in sternoclavicular joint septic arthritis. J Rheumatol 1989;16:413-4. [Google Scholar] |
| 8. | Pollack MS. Staphylococcal mediastinitis due to sternoclavicular pyarthrosis: CT appearance. J Comput Assist Tomogr 1990;14:924-7. [Google Scholar] |
| 9. | Chen WS, Wan YL, Lui CC, Lee TY, Wang KC. Extrapleural abscess secondary to infection of the sternoclavicular joint. Report of two cases. J Bone Joint Surg Am 1993;75:1835-9. [Google Scholar] |
| 10. | Crisostomo RA, Laskowski ER, Bond JR, Agerter DC. Septic sternoclavicular joint: A case report. Arch Phys Med Rehabil 2008;89:884-6. [Google Scholar] |
| 11. | Fordham S, Cope S, Sach M. Optimal management of sternoclavicular septic arthritis. Eur J Emerg Med 2009;16:219-20. [Google Scholar] |
| 12. | Abu Arab W, Khadragui I, Echavé V, Deshaies A, Sirois C, Sirois M. Surgical management of sternoclavicular joint infection. Eur J Cardiothorac Surg 2011;40:630-4. [Google Scholar] |
| 13. | Muesse JL, Blackmon SH, Ellsworth WA 4th, Kim MP. Treatment of sternoclavicular joint osteomyelitis with debridement and delayed resection with muscle flap coverage improves outcomes. Surg Res Pract 2014;2014:747315. [Google Scholar] |
| 14. | Murga A, Copeland H, Hargrove R, Wallen JM, Zaheer S. Treatment for sternoclavicular joint infections: A multi-institutional study. J Thorac Dis 2017;9:1503-8. [Google Scholar] |
| 15. | Yood RA, Goldenberg DL. Sternoclavicular joint arthritis. Arthritis Rheum 1980;23:232-9. [Google Scholar] |
Fulltext Views
5,356
PDF downloads
678





