Translate this page into:
Surgical management of a lumbar spine posterior element aneurysmal bone cyst in a child
2 Deprtment of Pediatric Orthopedic Surgery, King Abdul-Aziz Medical City, Riyadh, Saudi Arabia
Corresponding Author:
Abdulaziz M Bayounis
King Abdullah Specialized Children Hospital, Riyadh
Saudi Arabia
ba.younis@hotmail.com
| How to cite this article: Bayounis AM, Alsayegh SO, Almugren TS. Surgical management of a lumbar spine posterior element aneurysmal bone cyst in a child. J Musculoskelet Surg Res 2020;4:105-108 |
Abstract
The estimated annual incidence of aneurysmal bone cysts (ABCs) is 1.4 cases/100,000 people. Around 8%–30% of ABCs are located in the spine, which represents approximately 15% of all spinal tumors. Although ABCs are benign tumors, they can be locally aggressive and cause destruction of their hosting bones and pathological fractures. In spine ABC cases, compression of the spinal cord or instability of the spine might occur, and an urgent decision must be taken. We present a case of an 11-year-old boy who was found to have a second lumbar (L2) vertebral ABC and failed two sessions of sclerotherapy with a progression of the cyst that led to spinal compression. The decision was taken for surgical intervention with bone grafting and posterior instrumentation (L2–3) as there was instability due to facet destruction. We present a good outcome for surgical excision and short-segment fixation with a fusion of lumbar spine ABC in a patient who failed sclerotherapy in terms of cord decompression and a lower chance of recurrence.Introduction
Aneurysmal bone cyst (ABC) is defined by the World Health Organization as an expanding osteolytic lesion that consists of blood-filled spaces of variable size separated by connective tissue septa containing trabeculae of bone or osteoid tissue and giant osteoclast cells.[1] Although it is neither an aneurysm nor a bone cyst, it has been accepted throughout the world to be called ABC based on the radiological appearance.[2],[3] The estimated annual incidence of ABCs is 1.4 cases/100,000 people. It can be seen in any bone but is usually found in long bones' metaphysis.[4] Less than one-third of ABCs (8%–30%) are located in the spine, and they represent 15% of all spine tumors.[2],[4] Plain radiography, computed tomography (CT), and magnetic resonance imaging (MRI) are needed for diagnosing ABCs, and rarely open or imaging-guided needle biopsy is needed.[4],[5],[6]
In ABCs, therapeutic objectives that need to be solved other than the tumor itself are neurological and mechanical complications from the pressure effect. Although ABCs are benign tumors, they can be locally aggressive and cause destruction of their hosting bones and pathological fractures. In spine ABC cases, compression of the spinal cord or instability of the spine might occur, and an urgent decision must be taken.[1],[7] The standard of care for the medical treatment of ABC is embolization.[8] In this report, we present a good outcome for surgical excision and short-segment fixation with a fusion of lumbar spine ABC in a patient who failed sclerotherapy in terms of cord decompression and a lower chance of recurrence.
Case Report
An 11-year-old boy referred by his family physician to the pediatric orthopedic clinic as a case of lumbar spine lytic lesion at the level of the second lumbar (L2) vertebra. The patient recalls a history of a hit by a supermarket stroller 6 months before the presentation. He was cleared by an emergency physician through clinical examination and radiographs of the lumbar spine at the time of the injury. The pain continued in an on and off pattern after that incident until about 1 month before the presentation to our clinic. When the patient was seen by his family physician for follow-up, for a further medical assessment, as a bulge was noticed by the family. The patient was referred to the pediatric orthopedic clinic as a case of lower back pain with swelling. On examination, the patient had normal spinal curvature appearance with no deformities. There was a mild swelling over the L2 level with moderate tenderness. Sensory and motor neurological examinations were normal, with no upper motor neuron signs. Radiographs showed normal bone appearance with maintained vertebral heights and intervertebral disk spaces. The CT showed L2 posterior element expansile lytic lesion with no mass effect over the spinal cord [Figure - 1] and [Figure - 2]. Further investigation with MRI supported the diagnosis of an ABC with a fluid–fluid level containing bone lesion measuring 2.5 cm × 2.0 cm × 2.0 cm [Figure - 3]. The patient was referred to interventional radiology for two sessions of sclerotherapy with only 2 cc of doxycycline mixed with contrast and saline as there was a suspicion of communication to the spinal canal.
 |
| Figure 1: Anteroposterior and lateral lumbar spine X-ray |
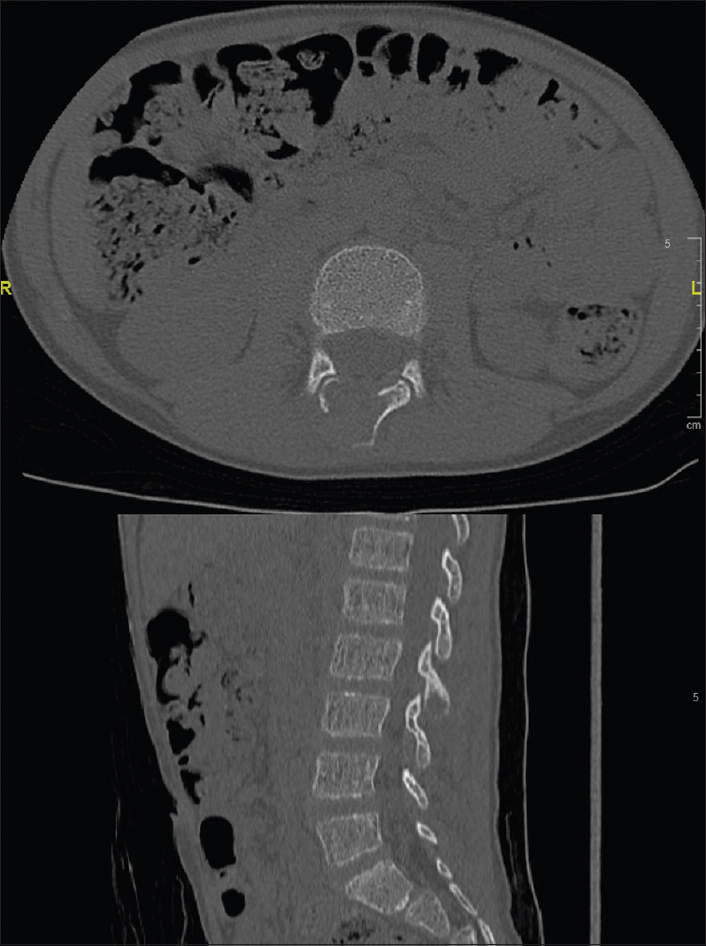 |
| Figure 2: Sagittal and coronal cuts of the lumbar spine computed tomography |
 |
| Figure 3: Sagittal and axial cuts of lumbar spine magnetic resonance imaging before sclerotherapy |
The patient returned to our clinic 1 month after the sclerotherapy for follow-up with progressing pain. On clinical examination, the size of the mass got larger with tenderness, and there was a right-sided L2 motor weakness “Graded 4/5.” Urgent MRI showed progression in the size of the L2 posterior element ABC measuring approximately 3.5 cm × 6 cm × 5.5 cm with the posterior intraspinal component, causing severe narrowing of the spinal canal and compression of cauda equina nerve roots [Figure - 4]. The plan was proceeding for surgical resection of the cyst with spinal instrumentation and fusion of L2–3. Our colleagues in interventional radiology were consulted and arranged an embolization session for the tumor 1 day before the surgery to decrease the intraoperative bleeding. A posterior spinal artery was seen under angiography arising from the right L1 segmental artery, and a dominant anterior spinal artery was arising from the left L2 segmental artery. Therefore, no embolization was performed at this level, considering the unfavorable risk to benefit ratio. Our colleagues in neurosurgery were consulted to join the procedure if a dural involvement was found intraoperatively.
 |
| Figure 4: Sagittal and axial cuts of lumbar spine magnetic resonance imaging after 2 sclerotherapy sessions |
Surgical procedure
Intraoperatively, the patient was intubated and positioned prone over a Jackson table with good padding to all bony prominences. A direct posterior approach for the lumbar spine was done reaching to the L2 level and exposing L1–3 levels, which showed a central mass with right-sided extension and muscle necrosis around. The cyst was peeled of the spinal canal and the dura. It was sent to the pathology laboratory in formalin labeled with the patient's name and medical record number [Figure - 5]. Hemostasis was achieved, followed by instrumentation of L2–3 with bone grafting for fusion, as the facet joints at the level of L2 and L3 were destructed by the bone cyst [Figure - 6]. Closure in layers after inserting a drain was done with sterile dressing followed by pressure dressing. A plaster body jacket cast was applied postoperatively for extra support to the fixation.
 |
| Figure 5: Mass removed from L2 lumbar spine |
 |
| Figure 6: Intraoperative fluoroscopy showing pedicle screws in L2–3 |
The patient mobilized on the 2nd postoperative day with normal neurovascular examinations, and the drain was removed on the 3rd day. At the 2-week follow-up visit in the clinic, the patient's wound was checked, which showed a clean healing wound. The pathology report confirmed the diagnosis of ABC. Six-week follow-up radiographs showed good implants' placement with no signs of cutout or failure. The plaster body jacket cast was removed 3 months after the surgery, and the repeated radiographs showed a good implants' placement with no signs of cutout or failure [Figure - 7]. The patient continued to have a regular follow-up in 6 months for 1 year with good alignment and fixation on the images. CT scan was done 18 months after the surgery, and there was no evidence of neither recurrence nor fixation failure [Figure - 8]. The patient and his family were happy with the final result, with no complaints.
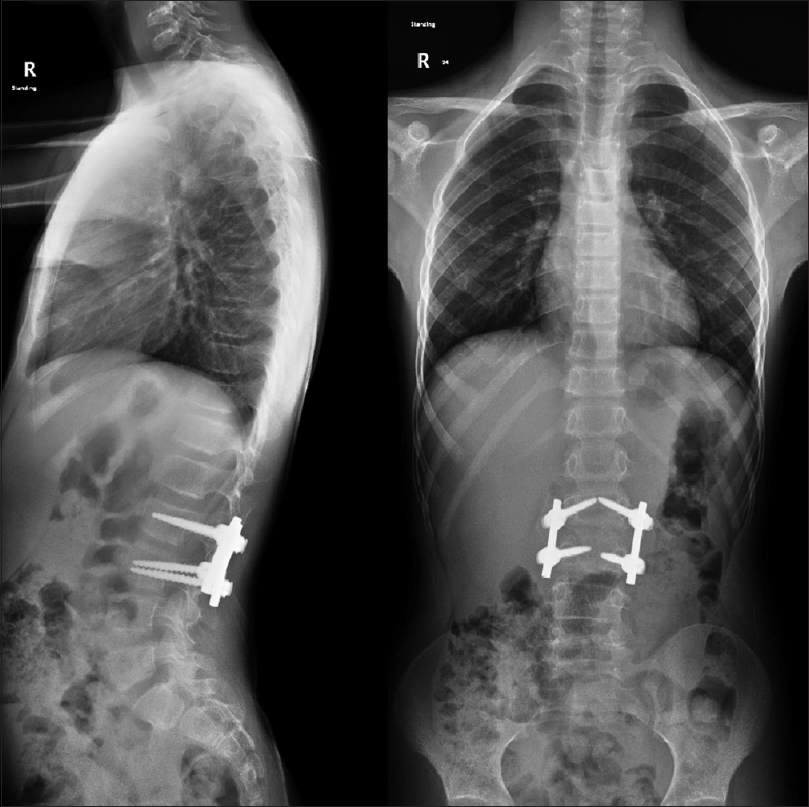 |
| Figure 7: Anteroposterior and lateral standing scoliosis series 6 months after the surgery |
 |
| Figure 8: Sagittal and coronal cuts of the lumbar spine computed tomography 18 months after the surgery |
Discussion
Spontaneous healing in lumbar spine ABC is rarely seen. The standard of care for the medical treatment of ABC is embolization.[8] Embolization can work in patients with no evidence of bone destruction responsible for spine deformity or neurological symptoms.[9],[10] Recurrence is usually seen in 10% of spine ABCs, and it appears in the first 6 months after the surgery.[4],[10] In our case, we found no evidence of recurrence for 18 months after the surgery. A study performed in France reported five cases of lumbar spine ABC treated surgically, and they were performing posterior instrumentation for four levels and more (two above and two below the lesion) not like our case in which we did two levels only.[11] Our instrumentation was limited to two levels and enhanced with a plaster body jacket cast that was removed 3 months after the surgery.
To minimize intraoperative blood loss, selective preoperative embolization is advised. Unfortunately, no embolization was performed in our case considering the unfavorable risk for spinal cord infarction as the posterior spinal artery was seen under angiography arising from the right L1 segmental artery, and a dominant anterior spinal artery was arising from the left L2 segmental artery. Decompression is recommended to be performed rapidly in cases of spinal cord involvement like our case. Stabilization of the spine must be completed by a bone graft with or without instrumentation. Postlaminectomy kyphosis is seen in about 37% of pediatric patients, as the compression of the cartilaginous endplates can cause wedging of the anterior vertebral bodies. During the adolescent growth phase, these deformities tend to progress rather than spontaneously correcting.[12] Fortunately, our patient had no deformities, as shown in the follow-up radiographs.
Although the standard medical care of ABC patients is embolization, a complete resection of ABCs is associated with a good prognosis in terms of decompression of the neural elements with a presenting risk of intraoperative bleeding. Preoperative tumor assessment and possible instrumentation planning is crucial for preventing possible postoperative spinal instability. In this case, we present a good outcome for surgical excision of a lumbar spine ABC with spinal cord decompression and a lower chance of recurrence. There is a limitation in this study as it is a single case, and further studies can be done.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. The patient's parents understand that his name and initials will not be published, and due efforts will be made to conceal his identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Authors' contributions
SOS was the primary surgeon, and he brought the case idea with logistic support. AMB and TSM did the literature review and contributed to the definition of intellectual content. AMB collected data and wrote initial and final drafts of the article. All authors have critically reviewed and approved the final draft and are responsible for the content and similarity index of the manuscript.
| 1. | Fletcher CD, Unni KK, Mertens F, editors. Pathology and Genetics of Tumours of Soft Tissue and Bone (IARC World Health Organization Classification of Tumours). 3rd ed. Lyons: IARC Press; 2006. p. 338-40. [Google Scholar] |
| 2. | de Kleuver M, van der Heul RO, Veraart BE. Aneurysmal bone cyst of the spine: 31 cases and the importance of the surgical approach. J Pediatr Orthop B 1998;7:286-92. [Google Scholar] |
| 3. | Bonakdarpour A, Levy WM, Aegerter E. Primary and secondary aneurysmal bone cyst: A radiological study of 75 cases. Radiology 1978;126:75-83. [Google Scholar] |
| 4. | Boriani S, de Iure F, Campanacci L, Gasbarrini A, Bandiera S, Biagini R, et al. Aneurysmal bone cyst of the mobile spine: Report on 41 cases. Spine (Phila Pa 1976) 2001;26:27-35. [Google Scholar] |
| 5. | Chan MS, Wong YC, Yuen MK, Lam D. Spinal aneurysmal bone cyst causing acute cord compression without vertebral collapse: CT and MRI findings. Pediatr Radiol 2002;32:601-4. [Google Scholar] |
| 6. | Mahnken AH, Nolte-Ernsting CC, Wildberger JE, Heussen N, Adam G, Wirtz DC, et al. Aneurysmal bone cyst: Value of MR imaging and conventional radiography. Eur Radiol 2003;13:1118-24. [Google Scholar] |
| 7. | Cottalorda J, Bourelle S. Modern concepts of primary aneurysmal bone cyst. Arch Orthop Trauma Surg 2007;127:105-14. [Google Scholar] |
| 8. | Kónya A, Szendröi M. Aneurysmal bone cysts treated by superselective embolization. Skeletal Radiol 1992;21:167-72. [Google Scholar] |
| 9. | Turker RJ, Mardjetko S, Lubicky J. Aneurysmal bone cysts of the spine: Excision and stabilization. J Pediatr Orthop 1998;18:209-13. [Google Scholar] |
| 10. | Papagelopoulos PJ, Currier BL, Shaughnessy WJ, Sim FH, Ebsersold MJ, Bond JR, et al. Aneurysmal bone cyst of the spine. Management and outcome. Spine (Phila Pa 1976) 1998;23:621-8. [Google Scholar] |
| 11. | Geffroy L, Hamel O, Odri GA, Guillard S, Passuti N, Gouin F, et al. Treatment of an aneurysmal bone cyst of the lumbar spine in children and teenagers, about five cases. J Pediatr Orthop B 2012;21:269-75. [Google Scholar] |
| 12. | Bell DF, Walker JL, O'Connor G, Tibshirani R. Spinal deformity after multiple-level cervical laminectomy in children. Spine (Phila Pa 1976) 1994;19:406-11. [Google Scholar] |
Fulltext Views
1,949
PDF downloads
322





