Translate this page into:
The efficacy of vein ensheathing in protecting peripheral nerve repair sites
2 Department of Hand Surgery, Queen Elizabeth Hospital, Birmingham, England, United Kingdom
Corresponding Author:
Rajive M Jose
Department of Hand Surgery, Queen Elizabeth Hospital, B15 2WB, Birmingham
United Kingdom
rajive.jose@uhb.nhs.uk
| How to cite this article: Thirumalai A, Jose RM, Power D. The efficacy of vein ensheathing in protecting peripheral nerve repair sites. J Musculoskelet Surg Res 2019;3:123-127 |
Abstract
Objectives: Peripheral nerve repairs can have a variable outcome depending on several factors. Neuroma in continuity at the repair site may limit functional recovery and is frequently associated with localised pain and sensitivity to mechanical stimulation. Extraneural scar may constrict the repair site, impeding axonal regeneration and resulting in adhesions to the nerve repair bed, reducing nerve glide and causing neurostenalgia. This study looked at the outcomes of using a segment of vein to ensheathe peripheral nerve repair sites in twenty patients to measure efficacy with validated functional outcomes and complications. Methods: This was a retrospective review of twenty cases performed in our unit between 2011 and 2015. Thirteen cases of vein ensheathing were performed to protect primary neurorrhaphy following traumatic lacerations without significant nerve loss. Seven cases were performed in secondary nerve repairs, of which five followed excision of a neuroma and two following neurolysis and repair. Results: There were 19 cases with a follow-up ranging from 24 to 72 months (mean of 32 months). One patient did not respond and was lost to follow-up. Twelve patients attended clinic for the long-term follow-up appointment and seven opted for a telephone evaluation. There were no clinical neuromata identified at the repair sites in the 12 patients attending the research clinic. Eighteen patients reported no scar hypersensitivity and 17 reported some sensory recovery following the repair. Two patients reported no sensory recovery following the nerve repair and 17 had diminished or protective sensations. Twelve patients had formal quantitative sensory testing with two-point discrimination of which two patients achieved only S3 (poor), nine patients achieved S3+ (good), and one achieved S4 (excellent) using the Mackinnon and Dellon classification of sensory recovery. Conclusion: Vein ensheathing is a useful technique which decreases the risk of symptomatic neuromas and adhesions following repair of sensory nerves in the upper limb. However, a randomised controlled trial will be needed to further validate the use of this technique.
Introduction
The outcome of peripheral nerve repair is influenced by many factors including the mechanism of trauma, the adequacy of debridement, time from injury, type of nerve, location of injury site, method of repair, tension across the repair site and the surgical bed. Neuroma in continuity at the repair site may limit functional recovery and is frequently associated with localised pain and sensitivity to mechanical stimulation. In a study, it was found that 3%–5% of patients with traumatic or iatrogenic peripheral nerve injury develop a painful neuroma, especially following trauma of small cutaneous sensory nerve branches.[1] The focus of the current research is to improve outcomes following nerve repair and include sutureless repair using adhesives, detensioning repair using entubulation within conduits, detensioning allograft interposition neurorrhaphy and modification of the repair site with adjunctive wraps to prevent axonal escape, reduce scar adhesion and support the repair site.[2]
The aim of this study is to describe the technique of vein ensheathing for the protection of neurorrhaphy and to measure efficacy with validated functional outcomes and complications.
Materials and Methods
The study was performed in a tertiary referral orthoplastic hand trauma unit. Between 2011 and 2015, twenty consecutive vein ensheathing procedures were recorded in a prospective database for the protection of neurorrhaphy sites. The mean age of participants was 37 years (range: 17–63). There were 18 male and 2 female participants. The anatomical distribution of the neurorrhaphy sites included 8 superficial radial nerves, 7 digital nerves, 3 dorsal branch ulnar nerves, 1 lateral cutaneous nerve of the forearm and 1 palmar cutaneous branch of the median nerve.
Thirteen cases of vein ensheathing were performed to protect primary neurorrhaphy following traumatic lacerations without significant nerve loss. Seven cases were performed in secondary nerve repairs, of which five followed excision of a neuroma and two following neurolysis and repair.
A nerve graft was used to bridge a gap in four cases where primary nerve repair was not possible, of which one in the case of a degloving injury and three in secondary nerve repairs following excision of a neuroma where a nerve gap of more than 20 mm dictated that a primary nerve repair could not be achieved without excessive tension. A single vein sheath was used to protect the neuro-anastomosis at both ends of the nerve graft.
All 13 patients who underwent exploration of traumatic lacerations had concomitant tendon injuries, which were repaired in addition to the primary microscope-assisted neurorrhaphy and vein ensheathing procedure.
The postoperative therapy protocol was defined by the associated non-nerve injury. Patients were assessed at 2 weeks, 6 weeks, and 6 months following nerve repair. Scar sensitivity, sensory recovery, pain, and complications were recorded at the final follow-up.
Patients were recalled for a 24-month outcome evaluation of the vein ensheathing neurorrhaphy after favuorable approvals from the Research Ethics Committee and the Health Research Authority. All 20 patients identified from the prospective database were invited by letter to attend a research clinic for further clinical assessment by a surgeon (first author). The sensitivity of the wound scar was assessed using a visual analogue scale. Tinel's sign at the repair site was used to determine whether there was a repair site neuroma. The sensibility of the affected area was assessed together with control areas from proximal to the site of injury and the same area on the contralateral limb. The innervation density was evaluated using static two-point discrimination. Patients unable to attend the research clinic were offered a telephone survey to record data for the study regarding scar sensitivity and qualitative information regarding the sensory recovery.
Surgical technique
Following regional or general anaesthesia, a pneumatic tourniquet is applied to the affected limb, which is exsanguinated, prepped, and draped. Using loupe magnification, the site of suspected nerve injury is explored and the proximal and distal nerve stumps are identified. Contused nerve ends are debrided using microsurgical instruments under an operating microscope. Mobilisation of both the proximal and distal nerve segments is performed to reduce the tension at the site of nerve repair. A superficial vein in the field of exploration is identified and lumen size is matched with the cut section of the nerve. A 2–3-cm segment of the superficial vein is harvested and both ends are tied off. Either the proximal or the distal end of the cut peripheral nerve is passed through the venous segment as a sleeve. The segment is selected based on the local anatomy and the ease of access for the nerve coaptation. End-to-end neurorrhaphy is performed under magnification using interrupted 9-0 nylon sutures. The venous sheath is then transposed to cover the neurorrhaphy site. If the vein sleeve is loose, then a single 9-0 nylon suture is used to secure the vein sheath to the epineurium [Figure - 1], [Figure - 2], [Figure - 3], [Figure - 4], [Figure - 5].
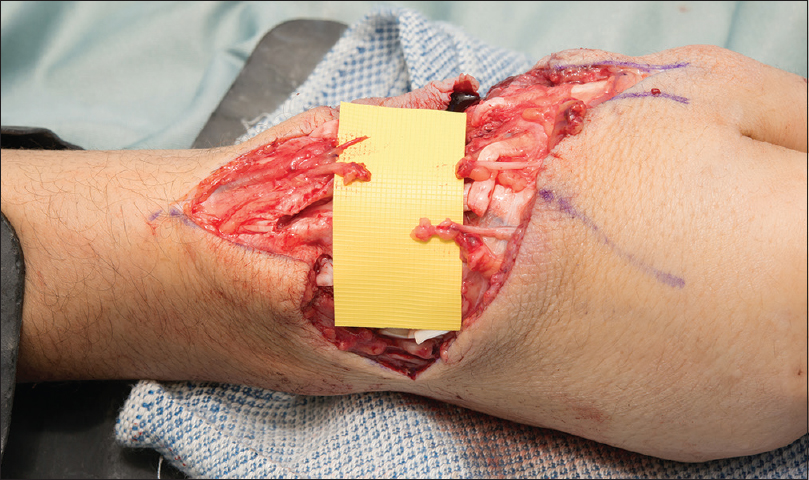 |
| Figure 1: Laceration over the dorsum of the hand with branches of superficial radial nerve injured |
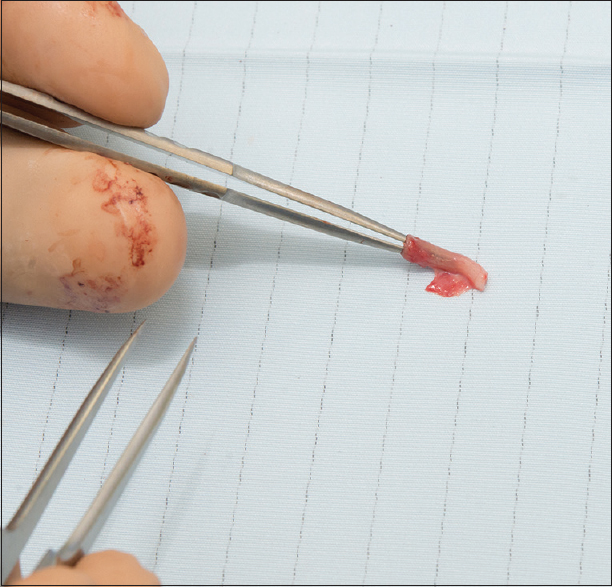 |
| Figure 2: A segment of the cephalic vein is harvested |
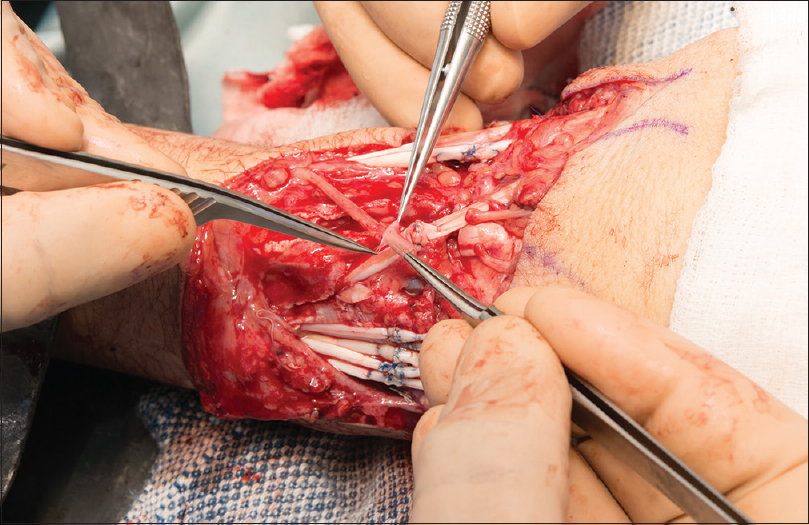 |
| Figure 3: The vein is threaded over the nerve end |
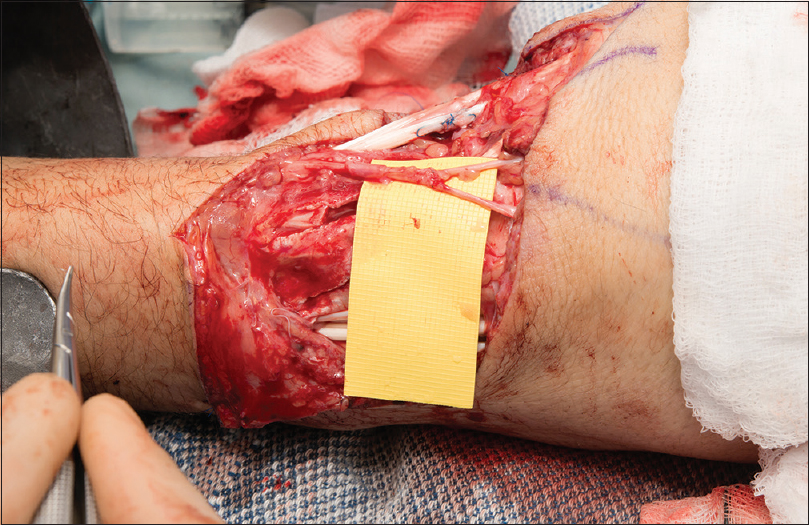 |
| Figure 4: Neurorrhaphy is complete |
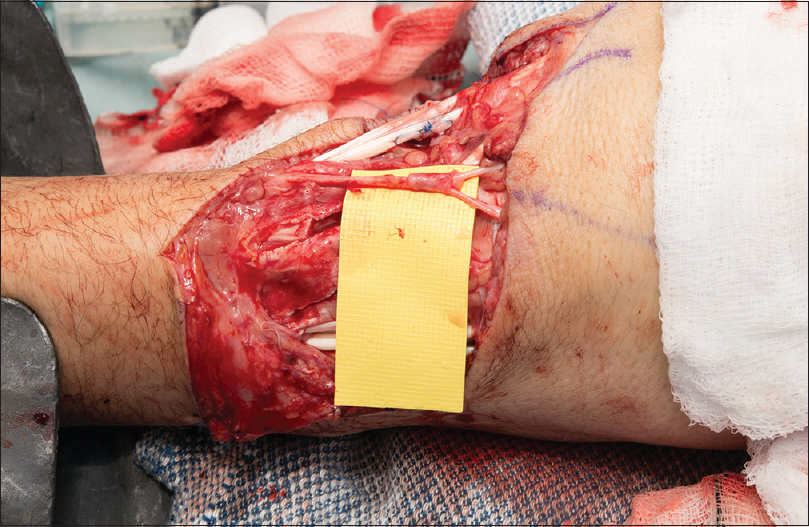 |
| Figure 5: Vein segment is pulled over the repair site |
In cases where there is a nerve gap that cannot be repaired directly without excessive tension, a nerve graft is placed to bridge the gap. The nerve graft is selected from a less critical sensory nerve in the same limb, typically the medial or lateral cutaneous nerves of the forearm or posterior interosseous nerve. A longer segment of the vein is harvested to ensheathe both sites of nerve repair with a single tube. If a long graft is used, a single vein tube can be transected to provide two individual cuffs that can be applied separately to the proximal and distal neurorrhaphy sites.
In cases where the nerve is in continuity, partial nerve injury requiring repair, or following secondary neurolysis, a longer segment of vein is harvested as above and opened up with a single longitudinal incision. The vein is then wrapped around the nerve segment and secured with 9-0 nylon. The intimal surface is in contact with the nerve. If it is a small nerve, the vein is used to completely ensheathe the nerve.
Results
Follow-up in 19 cases ranged from 24 to 72 months, with a mean of 32 months. One patient did not respond and was lost to follow-up. Twelve patients attended clinic for the long-term follow-up appointment and seven opted for a telephone discussion. The results are presented for the 12 cases which had objective assessment. The types of nerve repairs and the indications for vein ensheathing are represented in [Table - 1] and [Table - 2].


There were no clinical neuromata identified at the repair sites in the 12 patients attending the research clinic.
Two patients reported no sensory recovery following the nerve repair and 17 patients had diminished or protective sensations. Twelve patients have formal quantitative sensory testing with two-point discrimination and two patients achieved only S3 (poor), nine patients achieved S3+ (good) and one achieved S4 (excellent) using the Mackinnon and Dellon classification of sensory recovery.[3]
Of the patients who had phone interviews, six patients reported no scar hypersensitivity and all reported some sensory recovery following the repair. One patient remains under the care of pain services 5 years after a complex hand injury and reconstruction.
Discussion
More than 60% of peripheral nerve injuries occur in the upper limbs.[4] Repair and reconstruction of these peripheral sensory nerve injuries in the forearm and hand are challenging with frequent complications including scar sensitivity, neurostenalgia and neuroma formation as complications impacting on pain, functional recovery and quality of life.
Protection of the repair site from the surrounding tissues with the use of a biologically inert barrier may prevent axonal escape, nerve tether and potentially neuroma formation.
The ideal barrier should be biologically inert and protective without compression of the nerve repair site.[5] This is especially important for superficial sensory nerves where a neuroma can cause a sensitive cutaneous scar. The addition of a collagen tube to a repair site has demonstrated reduced pain at rest and pain on movement in a study comparing the outcome of nerve repairs in 101 patients undergoing digital replantation. There was no difference in sensory outcome.[4] A recent randomised study compared a control arm with 37 microsurgical digital nerve repairs with an interventional group with 37 nerve repairs plus augmentation with a chitosan nerve tube wrap. The results demonstrated improved sensory outcomes with less pain in the interventional group at 6 months. There was a symptomatic neuroma rate of 8% in the control group and none were reported in the intervention group. The criteria for diagnosis of a neuroma in the study were not reported. Neuroma formation may be influenced by extrinsic scar formation and repair site tether during mobilisation.[6]
Numerous conduits are available with evidence to support their use in short gap non-critical sensory peripheral nerve repair. A conduit used for the management of a nerve gap must maintain its cross-sectional area during digit mobilisation. The relative stiffness of a conduit lends itself to nerve gap management up to 15 mm or to be used as a detensioning tool leaving a 5-mm gap in simple transection injury; however, they are too rigid to be used as repair site adjuncts where a simple flexible barrier to scar is required. Flexible collagen nerve connectors are more suited to this application.[7] The cost of conduits and connectors is a barrier to widespread uptake in the absence of more robust data on their outcomes. Veins have been evaluated in nerve gap management; however, they failed to support axonal regeneration due to their flexibility and susceptibility to compression. Veins are readily available in the operative field, and the use of tubular vein grafts is an alternative autologous biological solution to provide support to a nerve repair site and prevent scar tether and axonal escape.[8]
Vein ensheathing of nerve repairs has been reported in the published literature with 16 cases of direct upper limb nerve repair plus superficial vein wrap procedures compared with 10 simple neurorrhaphies. The motor and sensory recovery was superior in the vein wrap group, and there were fewer pain complications.[8] It is more popular as a secondary procedure following neurolysis or neuroma excision where employment as an entubulation device for approximation of nerve gaps may assist nerve regeneration in short gaps. The entubulation technique does not produce reliable nerve regeneration in long gaps or in mobile digital segments where the lumen may become occluded during flexion.[8],[9],[10]
The limitations of this study include the variability of the nerves injured, the degree of injury, nerve tissue loss requiring grafting and associated injuries, which render functional scores of limited benefit. The study, therefore, has focused on scar sensitivity, symptomatic neuroma formation, pain, subjective sensory recovery and complications.
The sample size is too small and there is no comparator group to determine whether vein ensheathing confers an advantage in terms of nerve regeneration and sensory outcome after nerve repair. There is published evidence that supports the use of an adjunctive barrier at a repair site to reduce nerve tether and pain complications and this series supports that position.[4],[6] A study of motor nerve repair in rats has demonstrated no benefit in terms of motor regeneration when a bio-resorbable wrap was used around a microsurgical neurorrhaphy.[11] Whether using an adjunctive barrier as a support to the repair site reducing tension across the repair and potentially the number of sutures at the neurorrhaphy confers an advantage in terms of reducing neuroma formation and enhancing regeneration is beyond the scope of this study and will need to be evaluated in a well-constructed randomised controlled trial. There is no current evidence of sufficient power to support this hypothesis.
Wrapping a sensory nerve following neurolysis can prevent recurrence of adhesions. Alternatives to vein ensheathing in this scenario will be synthetic devices typically of collagen or polymer derivation[12],[13],[14] or biological wraps consisting of adipofascial flaps.[15],[16],[17]
The advantage of an adipofascial flap as a wrap around a nerve is that it will restore vascularity to the nerve bed and provide a membrane for functional nerve gliding. However, there is a cost to the patient with contour defects in the donor site affecting the cosmesis of this procedure.[17]
The ideal barrier should not incite an inflammatory response, should allow rapid revascularisation of the epineurium, and should act as a mechanical barrier to scar formation. There are several nerve wrapping products available: VIVOSORB® (Polyganics, Netherlands) – a bio-resorbable polycaprolactone sheet providing an inert physical barrier to scar for 10 weeks before hydrolysis and absorption over 12 months; AxoGuard® nerve protector (AxoGen Inc Alachua Florida, USA) – a porcine layered extracellular matrix collagen nerve protector allowing rapid vascular ingrowth and reconstitution of damaged epineurium[13] and Avive® processed human umbilical cord membrane (AxoGen Inc Alachua Florida, USA) – indicated for restoration of gliding surfaces adjacent to acutely injured nerves where the surgical bed and epineurium are damaged.[18] There is developing interest in using adjunctive barriers as delivery systems either for drugs that modify the inflammatory response or for stem cells to reduce fibrosis in repairing tissues or allow regeneration of tissues damaged or lost through trauma.
Although biological and synthetic materials are commercially available to protect nerves after nerve repair and neurolysis, these materials remain expensive, which limits their adoption, and in resource-poor health-care environments, the use of vein ensheathing remains a cost-effective solution to the issue of nerve repair site scar formation.
Conclusion
Scar formation around injured nerves results in poor nerve regeneration, impaired gliding, and neuropathic pain. In this study, vein ensheathing of injured nerves has resulted in useful sensory recovery, low rates of scar sensitivity, and no symptomatic neuromata formation. The technique is simple, acceptable to patients and cost-effective. A sufficiently powered comparative study will be required to demonstrate superior efficacy of commercial nerve barrier materials. Further modification of the inflammatory response may be possible by using these nerve repair adjunctive barrier wraps as drug or stem cell delivery systems.
Ethical consideration
Approval granted from Research Ethics Committee and the Health Research Authority for follow up of patients for research purposes.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Author's contributions
RMJ conceived and designed the study and wrote the initial draft, AT conducted research and collected and analyzed data. DP revised the article, write the discussion and provided logistic support. All authors have critically reviewed and approved the final draft and are responsible for the content and similarity index of the manuscript.
| 1. | Stokvis A, Coert JH. Operative treatment of painful neuromas. Ned Tijdschr Geneeskd 2011;155:A2592. [Google Scholar] |
| 2. | Barton MJ, Morley JW, Stoodley MA, Lauto A, Mahns DA. Nerve repair: Toward a sutureless approach. Neurosurg Rev 2014;37:585-95. [Google Scholar] |
| 3. | Wang Y, Sunitha M, Chung KC. How to measure outcomes of peripheral nerve surgery. Hand Clin 2013;29:349-61. [Google Scholar] |
| 4. | Zhu X, Wei H, Zhu H. Nerve wrap after end-to-end and tension-free neurorrhaphy attenuates neuropathic pain: A prospective study based on cohorts of digit replantation. Sci Rep 2018;8:620. [Google Scholar] |
| 5. | Adanali G, Verdi M, Tuncel A, Erdogan B, Kargi E. Effects of hyaluronic acid-carboxymethylcellulose membrane on extraneural adhesion formation and peripheral nerve regeneration. J Reconstr Microsurg 2003;19:29-36. [Google Scholar] |
| 6. | Neubrech F, Sauerbier M, Moll W, Seegmüller J, Heider S, Harhaus L, et al. Enhancing the outcome of traumatic sensory nerve lesions of the hand by additional use of a chitosan nerve tube in primary nerve repair: A randomized controlled bicentric trial. Plast Reconstr Surg 2018;142:415-24. [Google Scholar] |
| 7. | Ducic I, Fu R, Iorio ML. Innovative treatment of peripheral nerve injuries: Combined reconstructive concepts. Ann Plast Surg 2012;68:180-7. [Google Scholar] |
| 8. | Leuzzi S, Armenio A, Leone L, De Santis V, Di Turi A, Annoscia P, et al. Repair of peripheral nerve with vein wrapping. G Chir 2014;35:101-6. [Google Scholar] |
| 9. | Xu J, Varitimidis SE, Fisher KJ, Tomaino MM, Sotereanos DG. The effect of wrapping scarred nerves with autogenous vein graft to treat recurrent chronic nerve compression. J Hand Surg Am 2000;25:93-103. [Google Scholar] |
| 10. | Masear VR. Nerve wrapping. Foot Ankle Clin 2011;16:327-37. [Google Scholar] |
| 11. | Lee JY, Parisi TJ, Friedrich PF, Bishop AT, Shin AY. Does the addition of a nerve wrap to a motor nerve repair affect motor outcomes? Microsurgery 2014;34:562-7. [Google Scholar] |
| 12. | Kokkalis ZT, Mavrogenis AF, Ballas EG, Papagelopoulos PJ, Soucacos PN. Collagen nerve wrap for median nerve scarring. Orthopedics 2015;38:117-21. [Google Scholar] |
| 13. | Papatheodorou LK, Williams BG, Sotereanos DG. Preliminary results of recurrent cubital tunnel syndrome treated with neurolysis and porcine extracellular matrix nerve wrap. J Hand Surg Am 2015;40:987-92. [Google Scholar] |
| 14. | Lolley RD, Bose WJ, Bastian F, Bassam B, Meyer FN, Anderson LD, et al. Vein, silastic, and polyglycolic acid fine mesh: A comparative study in peripheral nerve repair. Ann Plast Surg 1995;35:266-71. [Google Scholar] |
| 15. | Yamamoto R, Motomiya M, Sakurai K, Sekiguchi H, Funakoshi T, Iwasaki N, et al. Application of free temporoparietal fascial flap for recurrent neural adhesion of superficial radial nerve – A case report. Microsurgery 2015;35:489-93. [Google Scholar] |
| 16. | Adani R, Tos P, Tarallo L, Corain M. Treatment of painful median nerve neuromas with radial and ulnar artery perforator adipofascial flaps. J Hand Surg Am 2014;39:721-7. [Google Scholar] |
| 17. | Samson D, Power DM. The adipofascial radial artery perforator flap: A Versatile reconstructive option in upper limb surgery. Hand Surg 2015;20:266-72. [Google Scholar] |
| 18. | Burman S, Tejwani S, Vemuganti GK, Gopinathan U, Sangwan VS. Ophthalmic applications of preserved human amniotic membrane: A review of current indications. Cell Tissue Bank 2004;5:161-75. [Google Scholar] |
Fulltext Views
2,506
PDF downloads
543





