Translate this page into:
Single-event multilevel surgery for crouching cerebral palsy children: Correlations with quality of life and functional mobility
2 Department of Rheumatology and Rehabilitation, Division of Pediatric Rehabilitation, Faculty of Medicine, Ain-Shams University, Cairo, Egypt
Corresponding Author:
John Amen
Department of Orthopedic Surgery, Division of Pediatric Orthopedics and Limb Reconstruction, Faculty of Medicine, Ain-Shams University, Cairo
Egypt
john_haleem@med.asu.edu.egTamer A. El-Sobky
Department of Orthopedic Surgery, Division of Pediatric Orthopedics and Limb Reconstruction, Faculty of Medicine, Ain-Shams University, Cairo
Egypt
tamer.ahmed@med.asu.edu.eg
| How to cite this article: Amen J, ElGebeily M, El-Mikkawy DM, Yousry AH, El-Sobky TA. Single-event multilevel surgery for crouching cerebral palsy children: Correlations with quality of life and functional mobility. J Musculoskelet Surg Res 2018;2:148-155 |
Abstract
Objectives: Crouch gait in cerebral palsy (CP) is characterized by excessive knee flexion throughout stance. Single-event multilevel orthopedic surgery is the standard of care to improve gait and function through correcting lever arm deformities in children with CP. A limited number of prospective studies on single-event multilevel orthopedic surgery for crouching CP children are available. We intended to evaluate the effectiveness of single-event multilevel surgery (SEMLS) regarding functional mobility, energy consumption during gait, and the mental, attitudinal, and lifestyle status of CP children with crouch gait on the short term. Methods: Thirty-four limbs in 18 children with bilateral spastic and crouching CP were enrolled prospectively and subjected to SEMLS. The mean age was 12 years (range, 5.5–18). Outcome measures included clinical couch examination parameters, walking speed, physiological cost index (PCI), functional mobility scale (FMS), and the World Health Organization Disability Assessment Schedule 2.0 (WHODAS 2.0). In addition, we used instrumented three-dimensional gait analysis as an outcome measure for some participants. Results: At a mean follow-up of 16 months (range, 12–22), the couch examination parameters, FMS at 5, 50, and 500 m, walking speed, PCI, and WHODAS 2.0 score showed a highly statistically significant improvement (P < 0.01). Conclusions: SEMLS for CP children and adolescents with crouch gait is effective for improving function, independence, energy consumption, and quality of life on the short term.
Introduction
Cerebral palsy (CP) is a major source of childhood disability globally. Motor retardation is an all-embracing feature among CP children.[1],[2],[3] While the primary neural insult is permanent, the secondary orthopedic manifestations are progressive, with ankle equinus and hip dislocation being two of the most prevalent deformities in CP children.[4],[5] Crouch gait is a prevalent gait pattern in ambulatory CP children. It is characterized by excessive knee flexion throughout stance, usually >15°.[5],[6],[7] In addition, deformities commonly occur at other levels such as excessive ankle dorsiflexion/mid-foot break and excessive hip flexion during stance. The pelvis is either in the normal sagittal alignment or tilted posteriorly or anteriorly.[5],[6] An important contributor to crouch gait is skeletal deformities causing lever arm dysfunction. These include increased femoral anteversion, external tibial torsion, ankle equinus, and foot deformities among others. Joint contractures also cause lever arm dysfunction as they prevent the adequate torque generation of muscles across joints and the optimal use of the ground reaction force in controlling joint movements.[6],[8] Surgical interventions for the treatment of equinus deformity that weaken the triceps surae complex and most importantly the soleus are another weighty cause of crouch gait. For example, over-lengthening of the triceps surae, an ill-timed Achilles tendon lengthening, or surgical management of equinus without addressing the proximal limb deformities can all precipitate or induce crouch gait. This is usually referred to as iatrogenic or surgeon-induced crouch.[5],[6] This occurs through impacting the knee extension-ankle planter flexion coupling mechanism.[6],[7] In addition, crouch gait may occur as a part of the natural history of gait progression in CP children, especially around puberty. This is usually attributed to the imbalance between body mass and muscle strength.[9]
Given that the cause of crouch gait is usually multifaceted and difficult to identify accurately, treatment is debatable. The dynamic deformities occurring with crouch gait are ideally remedied by conservative options, such as muscle-strengthening, spasticity reduction, and bracing.[10] Single-event multilevel surgery (SEMLS) may become necessary whenever fixed/static deformities ensue.[5],[6],[11] SEMLS is considered to be the mainstay of care to improve gait and function through correcting lever arm deformities. SEMLS allows for only one prime period of rehabilitation, reduces the need for repeated anesthetic sittings, and reduces hospital admissions.[12],[13],[14] Instrumented three-dimensional (3D) gait analysis (IGA) plays a supplementary role to aid clinical decision-making. It offers objective and quantifiable data in regard to kinematics, kinetics, and dynamic electromyography (EMG).[8]
Two large systematic reviews of literature reports on the use of SEMLS in crouching CP children revealed many shortcomings in the study methodologies of the original studies and research evidence extraction to inform decision-making.[5],[15] The objective physician-reported scales have been implemented at the expense of subjective patient/parent-reported quality of life scales, especially in orthopedic interventions in CP children. Furthermore, there was a paucity of prospective studies.[5],[15],[16] The first aim of this prospective study is to evaluate the effectiveness of SEMLS on the functional mobility of CP children with crouch gait on the short-term with the help of a validated scale. Second, it aims to assess the effect of SEMLS on energy consumption during gait. The third aim is to assess the responsiveness of the patients mental, attitudinal, and lifestyle status to orthopedic correction by SEMLS.
Materials and Methods
Demographics
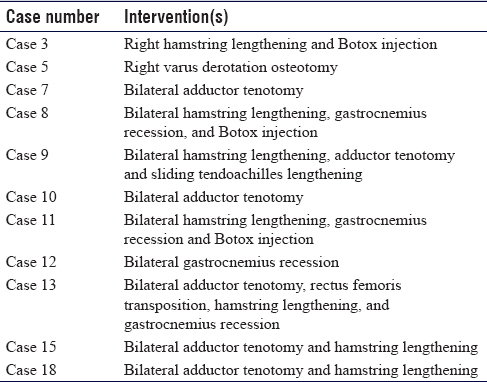
This institutional review board-approved prospective cohort study initially included 20 patients, who had bilateral spastic CP and walked with a crouch gait, who underwent SEMLS between March 2015 and October 2016. Of the 20 patients, two had lost follow-up, so the total number of patients who completed the study was 18 (34 limbs). Of the 18 patients, two underwent unilateral SEMLS. Ten patients (20 limbs) were males (59%) and eight patients (14 limbs) were females (41%). Mean age was 12 years (range, 5.5–18) and mean follow-up was 16 months (range, 12–22). According to the Gross Motor Function Classification System (GMFCS),[17] five (28%) patients were GMFCS II, ten (55%) were GMFCS III, and three (17%) were GMFCS IV. Two of the patients had an inborn error of metabolism, namely, well-controlled maple syrup urine disease.[18] Inclusion criteria were ambulatory children ≥5 years of age and adolescents with bilateral spastic CP and walked in a crouch and exhibited fixed joint deformities on couch examination. Crouch gait was defined as barefooted walking with knee flexion gait throughout the stance phase and crouch angle at the beginning of stance ≥15°, which may often be accompanied by hip flexion and ankle dorsiflexion. Exclusion criteria were botulinum toxin A injections within the preceding 6 months, orthopedic lower limb surgery within the preceding 12 months, noncommunicable patients. They were indicated for SEMLS if they experienced deterioration in gait implied by a reduction in speed or in walking distance or increased knee flexion in stance phase in the past year and/or pain, especially patellar pain and/or foot pain. In addition, bracing/footwear intolerance was considered an indication. The previous interventions are listed in [Table - 1].
Outcome measures
All patients received a standard orthopedic couch examination, including standard goniometric measurements of existing deformities. All goniometric measurements were performed by the same researcher to avoid inter-rater disagreement. We performed observational gait analysis with the patient barefooted and appropriately exposed. Walking speed and physiological cost index (PCI) were obtained in all patients according to the following equation: PCI (beats/m) = (final heart rate − resting heart rate)/speed of walking.
The functional mobility scale (FMS) was used for evaluation of the patients' mobility. FMS uses three distances (5, 50, and 500 m), which represent typical distances walked by children at home, at school, and in the community. For each distance, a rating of 1–6 was assigned depending on the amount of assistance required for mobility (6 being the highest ability).[19]
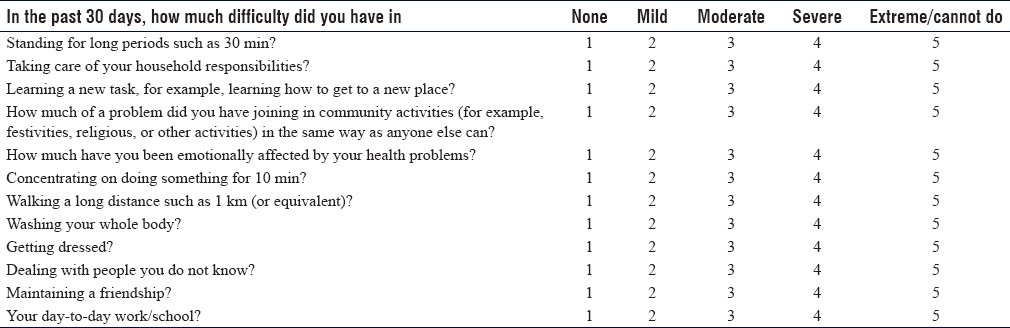
We used the World Health Organization Disability Assessment Schedule 2.0 (WHODAS 2.0) 12-item, interviewer-administered quality of life questionnaire to assess the mental, attitudinal, and lifestyle status of CP children.[20] When children or adolescents were unable to answer the questions independently, the parents or caregivers were asked to provide answers to the questionnaire [Table - 2]. Anteroposterior radiographs of the pelvis in a neutral hip rotation and lateral were taken to exclude hip migration, especially in children with low ambulatory capacity. We obtained dead lateral radiographs of the knee in flexion from 30° to 90° to measure the patellar height in accordance with Koshino index.[21]
IGA was done for 15 cases preoperative, nine of which did postoperative gait analysis as well. Kinematics, kinetics, and dynamic EMG were done in the same laboratory. However, kinetics was not done for patients walking with a frame as transmission of part of body weight though this walking aid may impact the validity of the joint kinetics. IGA was retaken after at least 1 year follow-up. We used Qualisys Track Manager QTM® Goteborg Sweden software to capture the data and visual 3D program to analyze data and create models. The markers necessary for analysis were set according to the modified lower limb model of Leardini that were tracked with eight Qualisys OQUS® infrared cameras and two video cameras, two AMTI portable force plates, and passive retroreflective markers.
Operative procedures for crouch gait
We defined SEMLS as two or more soft-tissue or bony surgical procedures at one or more anatomical levels. A total number of 146 index procedures were done to the 18 patients, 92 of which were soft tissue procedures and 54 were bony procedures [Table - 3]. All surgeries were performed by two authors (JA, TE). Supracondylar femoral extension osteotomy was done in concordance with Novacheck et al., 2009, except that we used thick K-wires for fixation.[22] Patellar tendon advancement was done according to Joseph et al.[23] Rectus femoris transfer to iliotibial band was performed according to Khouri and Desailly[24] We performed tailored medial hamstring release only. No lateral hamstring release was performed in this series. Gastrocnemius-soleus recession was done according to Vulpius technique.[25] Calcaneal-cuboid-cuneiform osteotomy was performed according to Rathjen and Mubarak.[26] We performed intramuscular recession of the tibialis posterior at the distal one-third of the medial leg and split tibialis anterior transfer to the cuboid.

Planning single-event multilevel surgery
A surgical plan for each patient was established by correlating the information of the couch examination, observational gait analysis, and occasionally radiography with the results of IGA. All patients were examined under general anesthesia (EUA) in the supine and prone position. Parameters of EUA were compared to preoperative couch examination to differentiate between spasticity and contractures, and surgical decisions were modified accordingly. The indications for SEMLS were dependent on the following general guidelines.
- Adductor tightness was defined as a limitation of hip abduction to ≤45° with extended knees. If the tightness persists in EUA, then all three adductors were released, namely the adductor longus, adductor brevis, and gracilis. If the tightness resolves partially in EUA, then the adductor longus and gracilis only were released
- Rectus femoris transposition was performed if two or more of the following criteria were met: (1) a positive rectus femoris stretch test on couch examination, (2) stiff knee gait as per the observational gait analysis, (3) kinematic data of reduced peak knee flexion during knee swing, and (4) kinematic data of delayed peak knee flexion during knee swing
- Hamstring tightness was indicted by a popliteal angle ≥40°. Hamstring tightness in the presence of posterior pelvic tilt was managed by either a medial hamstring release or femoral shortening in the context of a supracondylar extension osteotomy. Hamstring tightness in the presence of anterior pelvic tilt was managed conservatively
- Patellar tendon advancement was indicated either if there was an extension lag >10° on couch examination or if a supracondylar extension osteotomy was contemplated
- According to the Silfverskiold test, both gastrocnemius- induced and gastrocnemius-soleus-induced ankle equinus that persist in EUA were managed by a gastrocnemius-soleus recession. If ankle motion exceeds neutral dorsiflexion with an extended knee, no surgery was undertaken
- Fixed flexion deformity of the knee (FFDK) >10° that persists in EUA was managed by supracondylar extension osteotomy. Femoral shortening was added in FFDK of >30°. FFDK ≤10° can be subjected to minimal medial hamstring release with or without serial casting
- Internal femoral torsion was indicated if patellar squinting was evident during observational gait analysis, with an abnormal hip rotational profile in favor of an increased internal rotation range measured in the prone position.
Rehabilitation regimen
All patients received a combined surgical and rehabilitation program for the treatment of crouch gait. Rehabilitation of children with CP after SEMLS was tailored according to each patient condition. Patients were reassessed for all outcome measures postoperatively at regular follow-up intervals: 6 and 12 weeks, then three monthly during the 1st year, and 6 monthly thereafter.
Statistical analysis
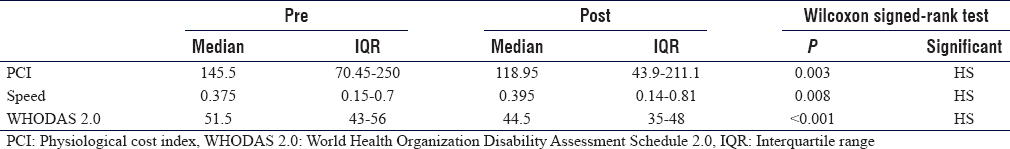
The collected data were revised, coded, tabulated, and introduced to a PC using the Statistical Package for the Social Science 20.0. Armonk, NY: IBM Corp. All parameters were measured preoperatively and at the final follow-up visit. The paired Wilcoxon signed-rank test was used to compare the preoperative and postoperative physical examination findings, walking speed, PCI, FMS, and WHODAS 2.0 score. The values were given as the median and the interquartile range. The significance level was defined by the probability (P value). The statistical significance was set at P < 0.05 (significance) and P < 0.01 (highly significant).
Results
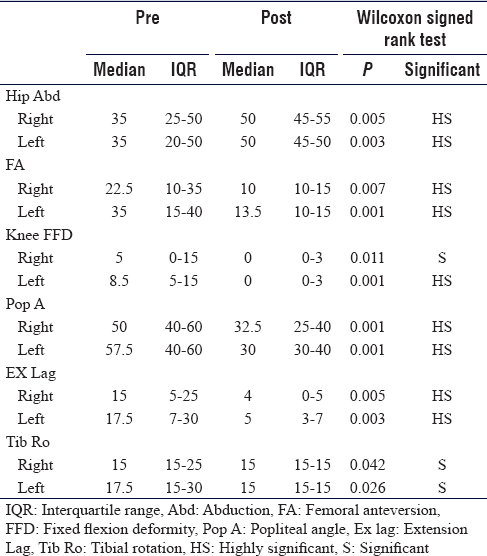
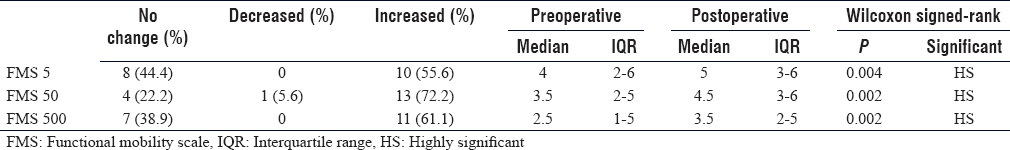
Generally, at the final follow-up visit, the values for the physical examination parameters, FMS, walking speed, PCI, and WHODAS 2.0, showed a highly statistically significant improvement [Table - 4], [Table - 5], [Table - 6]. The improvement in the measured values for IGA, namely stride, crouch angle at initial stance, and peak knee flexion in mid-swing, did not reach statistical significance [Figure - 1], [Figure - 2], [Figure - 3], [Figure - 4] and Video 1 shows improvements in gait parameters following SEMLS]. Complications included one superficial surgical site infection of patellar tendon advancement and one indolent low-grade infection in relation to the plate of the distal femoral osteotomy, presented by recurrent knee effusions. Both cases resolved following local wound care and plate removal 8 months postoperative. Tourniquet palsy developed in one patient. It resolved over a period of 6 months. Two patients needed additional surgery at 6 months postoperative in the form of patellar tendon advancement. This was secondary to deterioration of extension lag that was 5° preoperatively.
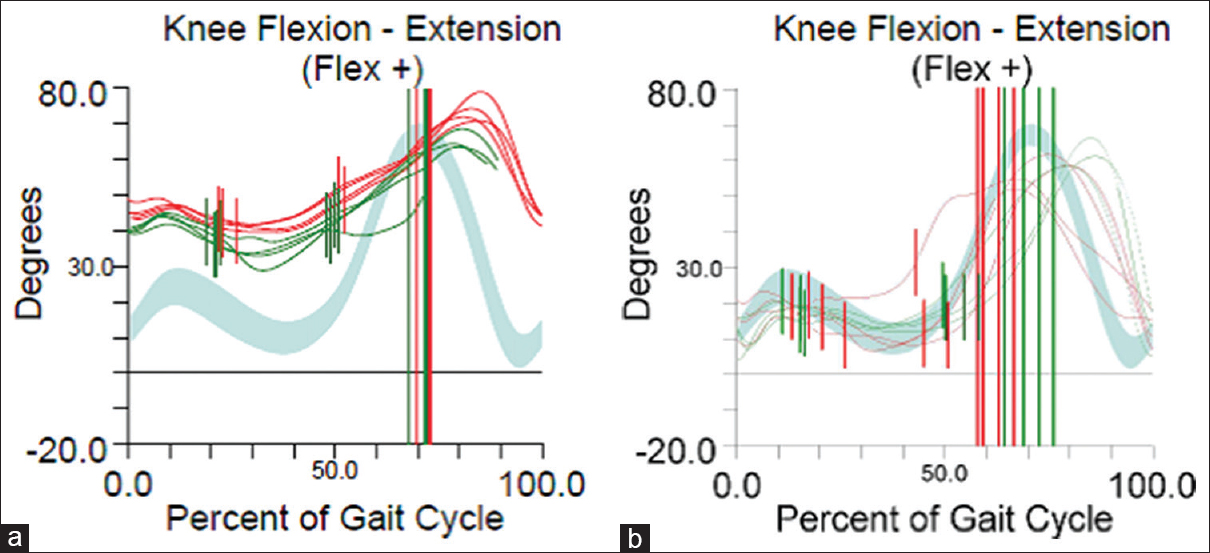 |
| Figure 1: Sagittal plane knee kinematics. (a) Preoperative data show an excessive crouch angle (knee flexion angle) at initial stance with a significantly delayed peak of knee flexion during swing (double bump sign). (b) Postoperative data show restoration of crouch angle and improved peak knee flexion in mid-swing and improved knee extension in late swing. Green = right, red = left, and grey represents the norm |
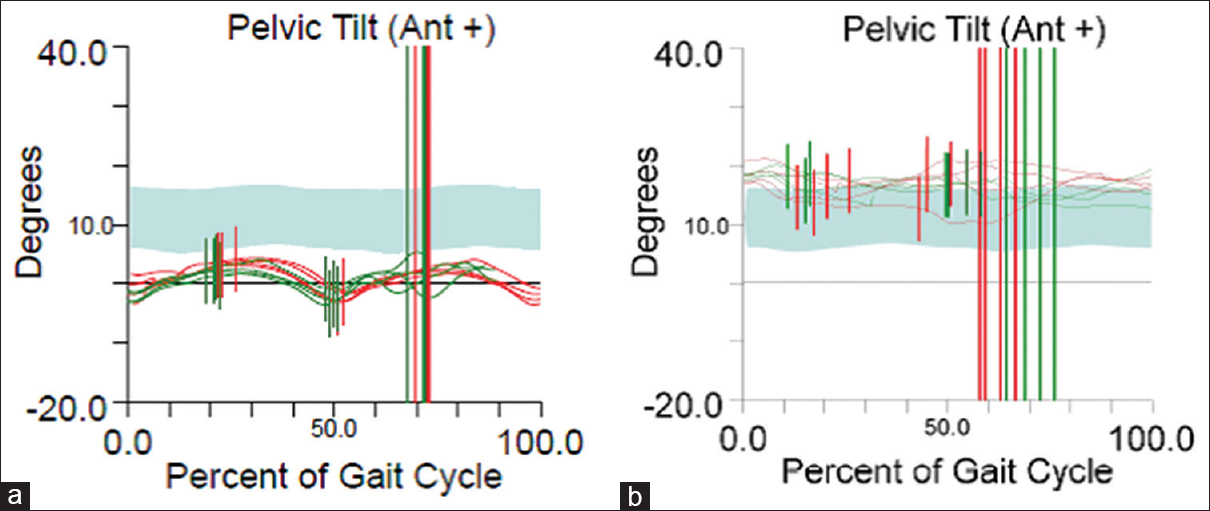 |
| Figure 2: Sagittal plane kinematics of the pelvis (same patient). (a) Preoperative data show a posterior pelvic tilt. (b) Postoperative data show restoration of the tilt |
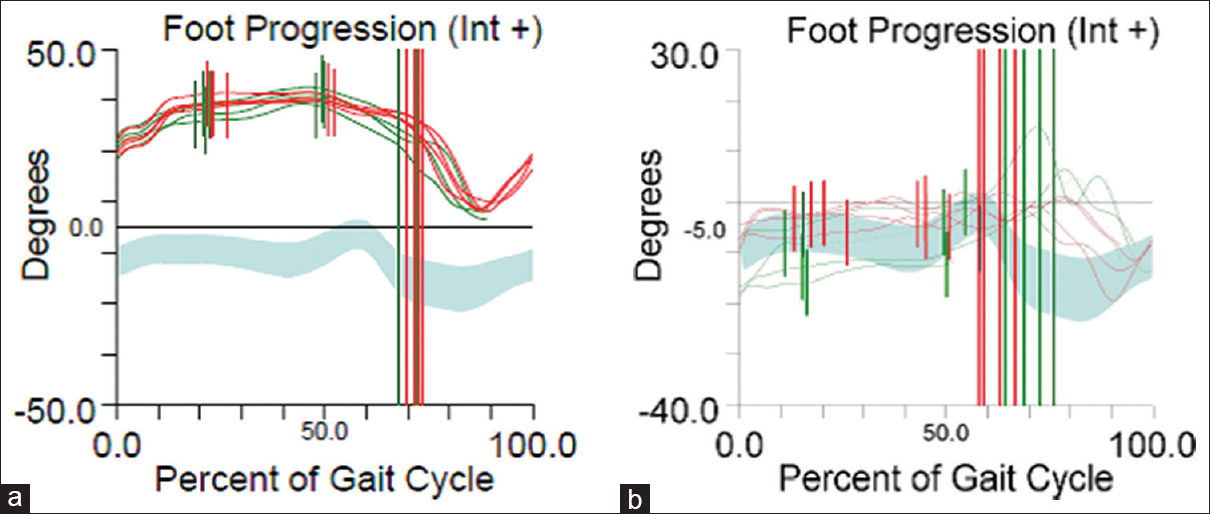 |
| Figure 3: Foot kinematics (same patient). (a) Preoperative data show a marked internal foot progression angle. (b) Postoperative shows restoration of foot progression angle |
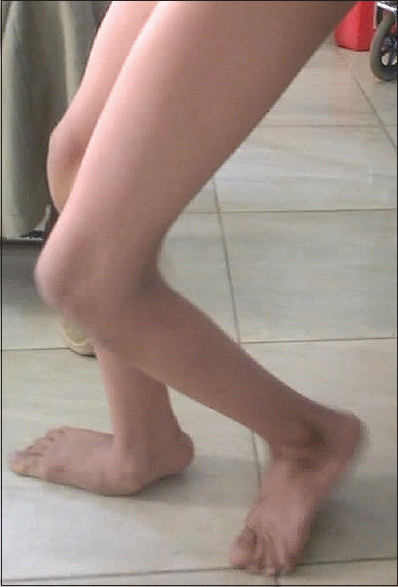 |
| Figure 4: A patient walking in crouch. Notice the dorsiflexion of the right foot arising from a severe mid-foot break. Mark the elevated right heel indicating a triceps surae contracture with valgus/pronated foot. Note the excessive flexion attitude of the left knee and hip which are in the pre/early swing phase of gait cycle |
Discussion
Crouch gait is an eventually disabling gait pattern in bilateral spastic CP children. The orthopedic management of crouching CP children is demanding because its exact pathomechanics has not been fully unraveled. In addition, the multiplanar and multilevel nature of skeletal deformities and the variability in patients' clinical profiles create decision-making difficulties with regard to the choice and timing of the surgical procedures.[6],[27] The presence of a wide array of outcome instruments used to evaluate the performance SEMLS may also complicate the interpretation of the results and impact the generalization of the conclusions.[5],[11],[12],[28]
Several authors have employed FMS to assess the outcome of SEMLS in spastic CP children.[19],[29] Generally, they noticed a significant improvement of FMS by 2 years postoperative.[19],[29] Particularly, Terjesen et al. noticed that this improvement was maintained at 5 years postoperative.[29] In our study, we reported a similar improvement in the FMS on the short term. This corroborates the role of FMS as an important outcome measure in SEMLS for ambulatory CP children. Contrastingly, the GMFCS is mainly used as a categorization tool.[5] In our study, the walking speed and PCI were used as a measure of energy consumption. Both showed significant improvement at the final follow-up.
Recently, the use of quality of life measures in CP children[30],[31],[32],[33],[34] and their caregivers.[34],[35],[36],[37] has become increasingly popular. The use of quality of life measures aims at assessing how they relate to impairments in CP children.[31],[32] Besides, they can be used as an outcome measure for health-related interventions.[35],[36],[37],[38] We used the WHODAS 2.0 questionnaire, 12-item interviewer-administered version to assess the responsiveness of the patients' mental, attitudinal, and lifestyle status to orthopedic correction by SEMLS. The WHODAS 2.0 has the advantages of being a short and simple instrument that can measure health and disability in clinical practice. It can produce standardized disability levels and profiles and is directly linked at the level of the concepts to the International Classification of Functioning, Disability and Health. To our knowledge, this is the first study to use this score in CP children. Our reported values at the short-term follow-up showed significant improvements. Studies using quality of life measures reported similar improvements in CP children and their caregivers following multilevel surgery[36],[37],[38] and major spine surgery.[35]
Evidence suggests that gait improvements following SEMLS are durable on the intermediate[14],[29] and on the long term.[5],[12],[39],[40] Nevertheless, the notion of SEMLS with a subsequent single rehabilitation regimen has been challenged.[12],[29],[41],[42],[43] The previous authors reported the need for additional surgeries as early as 1-year postoperative and in up to 65% of the study participants in some series.[12],[29],[41],[42],[43] The reasons for additional surgery varied from the emergence of new deformities to relapsed deformities that had been promptly corrected at index surgery, with some being minor in nature.[12],[29],[41],[42],[43] In our series, we needed additional surgeries in four patients (22%) because of the occurrence of new deformities that were nonexistent at the time of index surgery. The findings of our study conform to the observations of the previous authors. SEMLS is thus an inaccurate nomenclature. A “multilevel surgery” is a more representative term of orthopedic interventions performed on crouching CP children.[12],[14] In that regard, various studies advocate strict adherence to clinical assessment principles including observational gait assessment as a cost-effective way to reduce the needed for additional surgeries, improve decision-making, and improve outcome evaluation.[42],[43],[44] It seems plausible that we did not undergo any bony hip reconstruction because hip dislocation is usually linked to children with low ambulatory capacity, especially GMFCS V.[45],[46] Likewise, the limited use of intramuscular psoas lengthening in our series – 5% of operated limbs – goes in line with recent studies, which demonstrated that it does not yield additional biomechanical gains.[47],[48]
Strengths and weaknesses
The strength of this study lies in its prospective nature and its multidisciplinary team. This is because retrospective studies usually have imperfect control over bias and confounding variables.[4] The diversity of the outcome measures used is another source of study strength. Yet, our study had certain weaknesses. The sample size was relatively small. The follow-up period was short alike. Although the WHODAS 2.0 questionnaire used in our study can measure health and disability in clinical practice, we have not specifically tested its reliability and validity for CP children. Pain is one of the most noteworthy symptoms in CP children and adolescents.[49],[50] Yet, it is frequently underreported by researchers. Generally, validated and reliable assessment tools for pain are lacking.[51] We did not use a pain assessment tool in our study. We consider this a study limitation because the negative impact of pain can undercut the quality of life and motor capabilities of CP children.
Conclusion
- SEMLS for CP children with crouch gait is associated with significant gains in functional mobility on the short term
- SEMLS impacts the quality of life of crouching CP children positively as per the WHODAS 2.0 on the short term
- This study overemphasizes the role of a comprehensive management approach to crouch gait, namely the orthopedic surgery and the postoperative rehabilitation regimen
- In the decision-making process and postoperative follow-up alike, IGA was supplementary to the clinical tools. Regardless of its perceived efficiency, IGA is no substitute for a comprehensive clinical assessment
- This study suggests that using the term “multilevel surgery” instead of “SEMLS” may be more practical to describe the orthopedic intervention for crouching CP children.
Recommendations
- CP researchers are encouraged to conduct “prospective” studies that allow for bias control and allow for reporting of all actually and possibly relevant data
- CP researchers are encouraged to integrate validated quality of life scales into the outcome measures used for SEMLS
- Because lack of adherence by patients to the hospital-centered rehabilitation protocols might impact the outcomes of surgery, the role of family-centered care should be expanded.
Ethical approval and declaration of patient consent
The authors of this institutional review board-approved study certify that they have obtained all appropriate patient consent forms. In the form, the parents have given their consent for their children's images and other clinical information to be reported in the journal. The parents understand that names and initials of the patients will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Acknowledgment
We are grateful to Dr. Mai Abdelmohsen Ahmed for performing the instrumented three-dimensional gait analysis.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Authors' contributions
TAE and JA conceived and designed the study. Otherwise, all authors contributed equally to the acquisition, analysis, and interpretation of data. All authors have reviewed the study critically for important intellectual content, have checked the article for plagiarism, and are responsible for the content and similarity index of the manuscript. Finally, all authors approved the version to be published.
| 1. | Gowda VK, Kumar A, Shivappa SK, Srikanteswara PK, Shivananda, Mahadeviah MS, et al. Clinical profile, predisposing factors, and associated co-morbidities of children with cerebral palsy in South India. J Pediatr Neurosci 2015;10:108-13. [Google Scholar] |
| 2. | Colver A, Fairhurst C, Pharoah PO. Cerebral palsy. Lancet 2014;383:1240-9. [Google Scholar] |
| 3. | El-Tallawy HN, Farghaly WM, Shehata GA, Rageh TA, Metwally NA, Badry R, et al. Cerebral palsy in Al-Quseir City, Egypt: Prevalence, subtypes, and risk factors. Neuropsychiatr Dis Treat 2014;10:1267-72. [Google Scholar] |
| 4. | El-Sobky TA, Fayyad TA, Kotb AM, Kaldas B. Bony reconstruction of hip in cerebral palsy children gross motor function classification system levels III to V: A systematic review. J Pediatr Orthop B 2018;27:221-30. [Google Scholar] |
| 5. | Galey SA, Lerner ZF, Bulea TC, Zimbler S, Damiano DL. Effectiveness of surgical and non-surgical management of crouch gait in cerebral palsy: A systematic review. Gait Posture 2017;54:93-105. [Google Scholar] |
| 6. | Kedem P, Scher DM. Evaluation and management of crouch gait. Curr Opin Pediatr 2016;28:55-9. [Google Scholar] |
| 7. | Vuillermin C, Rodda J, Rutz E, Shore BJ, Smith K, Graham HK, et al. Severe crouch gait in spastic diplegia can be prevented: A population-based study. J Bone Joint Surg Br 2011;93:1670-5. [Google Scholar] |
| 8. | Theologis T. Lever arm dysfunction in cerebral palsy gait. J Child Orthop 2013;7:379-82. [Google Scholar] |
| 9. | Rethlefsen SA, Blumstein G, Kay RM, Dorey F, Wren TA. Prevalence of specific gait abnormalities in children with cerebral palsy revisited: Influence of age, prior surgery, and gross motor function classification system level. Dev Med Child Neurol 2017;59:79-88. [Google Scholar] |
| 10. | Ryan JM, Cassidy EE, Noorduyn SG, O'Connell NE. Exercise interventions for cerebral palsy. Cochrane Database Syst Rev 2017;6:CD011660. [Google Scholar] |
| 11. | Kim HY, Cha YH, Byun JY, Chun YS, Choy WS. Changes in gait parameters after femoral derotational osteotomy in cerebral palsy patients with medial femoral torsion. J Pediatr Orthop B 2018;27:194-9. [Google Scholar] |
| 12. | Dreher T, Thomason P, Švehlík M, Döderlein L, Wolf SI, Putz C, et al. Long-term development of gait after multilevel surgery in children with cerebral palsy: A multicentre cohort study. Dev Med Child Neurol 2018;60:88-93. [Google Scholar] |
| 13. | Lamberts RP, Burger M, du Toit J, Langerak NG. A systematic review of the effects of single-event multilevel surgery on gait parameters in children with spastic cerebral palsy. PLoS One 2016;11:e0164686. [Google Scholar] |
| 14. | Thomason P, Selber P, Graham HK. Single event multilevel surgery in children with bilateral spastic cerebral palsy: A 5 year prospective cohort study. Gait Posture 2013;37:23-8. [Google Scholar] |
| 15. | McGinley JL, Dobson F, Ganeshalingam R, Shore BJ, Rutz E, Graham HK, et al. Single-event multilevel surgery for children with cerebral palsy: A systematic review. Dev Med Child Neurol 2012;54:117-28. [Google Scholar] |
| 16. | El-Sobky TA, Mahran M. Comment on Silverio, et al: Proximal femur prosthetic interposition arthroplasty for painful dislocated hips in children with cerebral palsy. J Child Orthop 2017;11:237-8. [Google Scholar] |
| 17. | Palisano RJ, Rosenbaum P, Bartlett D, Livingston MH. Content validity of the expanded and revised gross motor function classification system. Dev Med Child Neurol 2008;50:744-50. [Google Scholar] |
| 18. | Huntsman R, Lemire E, Norton J, Dzus A, Blakley P, Hasal S, et al. The differential diagnosis of spastic diplegia. Arch Dis Child 2015;100:500-4. [Google Scholar] |
| 19. | Harvey A, Graham HK, Morris ME, Baker R, Wolfe R. The functional mobility scale: Ability to detect change following single event multilevel surgery. Dev Med Child Neurol 2007;49:603-7. [Google Scholar] |
| 20. | World Health Organization. Manual for WHO Disability Assessment Schedule – WHODAS 2.0. World Health Organization; 2010. Available from: http://www.who.int/icidh/whodas/. [Last accessed on 2018 Feb 23]. [Google Scholar] |
| 21. | Koshino T, Sugimoto K. New measurement of patellar height in the knees of children using the epiphyseal line midpoint. J Pediatr Orthop 1989;9:216-8. [Google Scholar] |
| 22. | Novacheck TF, Stout JL, Gage JR, Schwartz MH. Distal femoral extension osteotomy and patellar tendon advancement to treat persistent crouch gait in cerebral palsy. Surgical technique. J Bone Joint Surg Am 2009;91 Suppl 2:271-86. [Google Scholar] |
| 23. | Joseph B, Reddy K, Varghese RA, Shah H, Doddabasappa SN. Management of severe crouch gait in children and adolescents with cerebral palsy. J Pediatr Orthop 2010;30:832-9. [Google Scholar] |
| 24. | Khouri N, Desailly E. Rectus femoris transfer in multilevel surgery: Technical details and gait outcome assessment in cerebral palsy patients. Orthop Traumatol Surg Res 2013;99:333-40. [Google Scholar] |
| 25. | Tinney A, Thomason P, Sangeux M, Khot A, Graham HK. The transverse Vulpius gastrocsoleus recession for equinus gait in children with cerebral palsy. Bone Joint J 2015;97-B:564-71. [Google Scholar] |
| 26. | Rathjen KE, Mubarak SJ. Calcaneal-cuboid-cuneiform osteotomy for the correction of valgus foot deformities in children. J Pediatr Orthop 1998;18:775-82. [Google Scholar] |
| 27. | Wilson NC, Chong J, Mackey AH, Stott NS. Reported outcomes of lower limb orthopaedic surgery in children and adolescents with cerebral palsy: A mapping review. Dev Med Child Neurol 2014;56:808-14. [Google Scholar] |
| 28. | Nicholson K, Lennon N, Church C, Miller F. Gait analysis parameters and walking activity pre- and postoperatively in children with cerebral palsy. Pediatr Phys Ther 2018;30:203-7. [Google Scholar] |
| 29. | Terjesen T, Lofterød B, Skaaret I. Gait improvement surgery in ambulatory children with diplegic cerebral palsy. Acta Orthop 2015;86:511-7. [Google Scholar] |
| 30. | Power R, King C, Muhit M, Heanoy E, Galea C, Jones C, et al. Health-related quality of life of children and adolescents with cerebral palsy in low- and middle-income countries: A systematic review. Dev Med Child Neurol 2018;60:469-79. [Google Scholar] |
| 31. | Das S, Aggarwal A, Roy S, Kumar P. Quality of life in Indian children with cerebral palsy using cerebral palsy-quality of life questionnaire. J Pediatr Neurosci 2017;12:251-4. [Google Scholar] |
| 32. | Colver A, Rapp M, Eisemann N, Ehlinger V, Thyen U, Dickinson HO, et al. Self-reported quality of life of adolescents with cerebral palsy: A cross-sectional and longitudinal analysis. Lancet 2015;385:705-16. [Google Scholar] |
| 33. | Gilson KM, Davis E, Reddihough D, Graham K, Waters E. Quality of life in children with cerebral palsy: Implications for practice. J Child Neurol 2014;29:1134-40. [Google Scholar] |
| 34. | Dobhal M, Juneja M, Jain R, Sairam S, Thiagarajan D. Health-related quality of life in children with cerebral palsy and their families. Indian Pediatr 2014;51:385-7. [Google Scholar] |
| 35. | Jain A, Sullivan BT, Shah SA, Samdani AF, Yaszay B, Marks MC, et al. Caregiver perceptions and health-related quality-of-life changes in cerebral palsy patients after spinal arthrodesis. Spine (Phila Pa 1976) 2018;43:1052-6. [Google Scholar] |
| 36. | Stephan-Carlier A, Facione J, Speyer E, Rumilly E, Paysant J. Quality of life and satisfaction after multilevel surgery in cerebral palsy: Confronting the experience of children and their parents. Ann Phys Rehabil Med 2014;57:640-52. [Google Scholar] |
| 37. | Lee KM, Chung CY, Park MS, Lee SH, Choi IH, Cho TJ, et al. Level of improvement determined by PODCI is related to parental satisfaction after single-event multilevel surgery in children with cerebral palsy. J Pediatr Orthop 2010;30:396-402. [Google Scholar] |
| 38. | Himpens E, Franki I, Geerts D, Tack R, Van der Looven R, Van den Broeck C, et al. Quality of life in youngsters with cerebral palsy after single-event multilevel surgery. Eur J Paediatr Neurol 2013;17:401-6. [Google Scholar] |
| 39. | Švehlík M, Steinwender G, Lehmann T, Kraus T. Predictors of outcome after single-event multilevel surgery in children with cerebral palsy: A retrospective ten-year follow-up study. Bone Joint J 2016;98-B: 278-81. [Google Scholar] |
| 40. | Yu S, Rethlefsen SA, Wren TA, Kay RM. Long-term ambulatory change after lower extremity orthopaedic surgery in children with cerebral palsy: A retrospective review. J Pediatr Orthop 2015;35:285-9. [Google Scholar] |
| 41. | Rutz E, Baker R, Tirosh O, Brunner R. Are results after single-event multilevel surgery in cerebral palsy durable? Clin Orthop Relat Res 2013;471:1028-38. [Google Scholar] |
| 42. | Wren TA, Kalisvaart MM, Ghatan CE, Rethlefsen SA, Hara R, Sheng M, et al. Effects of preoperative gait analysis on costs and amount of surgery. J Pediatr Orthop 2009;29:558-63. [Google Scholar] |
| 43. | Gough M, Shortland AP. Can clinical gait analysis guide the management of ambulant children with bilateral spastic cerebral palsy? J Pediatr Orthop 2008;28:879-83. [Google Scholar] |
| 44. | Khouri N, Desailly E. Contribution of clinical gait analysis to single-event multi-level surgery in children with cerebral palsy. Orthop Traumatol Surg Res 2017;103:S105-11. [Google Scholar] |
| 45. | Dohin B. The spastic hip in children and adolescents. Orthop Traumatol Surg Res 2018. pii: S1877-0568(18)30197-X. [Google Scholar] |
| 46. | Klochkova OA, Kurenkov AL, Kenis VM. Development of contractures in spastic forms of cerebral palsy: Pathogenesis and prevention. Pediatr Traumatol Orthopaed Reconstr Surg 2018;6:58-66. [Google Scholar] |
| 47. | Mallet C, Simon AL, Ilharreborde B, Presedo A, Mazda K, Penneçot GF, et al. Intramuscular psoas lengthening during single-event multi-level surgery fails to improve hip dynamics in children with spastic diplegia. Clinical and kinematic outcomes in the short- and medium-terms. Orthop Traumatol Surg Res 2016;102:501-6. [Google Scholar] |
| 48. | Rutz E, Gaston MS, Tirosh O, Brunner R. Hip flexion deformity improves without psoas-lengthening after surgical correction of fixed knee flexion deformity in spastic diplegia. Hip Int 2012;22:379-86. [Google Scholar] |
| 49. | Westbom L, Rimstedt A, Nordmark E. Assessments of pain in children and adolescents with cerebral palsy: A retrospective population-based registry study. Dev Med Child Neurol 2017;59:858-63. [Google Scholar] |
| 50. | Alriksson-Schmidt A, Hägglund G. Pain in children and adolescents with cerebral palsy: A population-based registry study. Acta Paediatr 2016;105:665-70. [Google Scholar] |
| 51. | Kingsnorth S, Orava T, Provvidenza C, Adler E, Ami N, Gresley-Jones T, et al. Chronic pain assessment tools for cerebral palsy: A systematic review. Pediatrics 2015;136:e947-60. [Google Scholar] |
Fulltext Views
5,464
PDF downloads
580





